
Concept explainers
Interpretation:
A structure for the alcohol with molecular formula C9H12O and the following spectral data is to be proposed.
1HNMR: 0.88 δ (triplet,Rel.area=3.00); 1.80 δ (quintet, Rel.area=2.00); 2.32 δ (Rel.area=1.00); 4.54 δ (triplet, Rel.area=1.00); 7.24 δ (Rel.area=5.00).
Concept introduction:
In 1HNMR spectrum, the alcoholic proton absorption occurs in the range 3.4 δ - 4.5 δ while that of phenolic proton occur in the range 3.0 to 8.0. The
The absorption due to 10 alkyl group (CH3) is seen around 0.7 δ - 1.3 δ, that due to a 20 alkyl group (CH2) is seen around 1.2 δ - 1.6 δ while that due to 30 alkyl group (CH) is seen around 1.4 δ - 1.8 δ The multiplicity of a signal gives an idea about the protons present in the adjacent carbons.
To propose:
A structure for the alcohol with molecular formula C9H12O and the following spectral data is to be proposed.
1HNMR: 0.88 δ (triplet,Rel.area=3.00); 1.80 δ (quintet, Rel.area=2.00); 2.32 δ (Rel.area=1.00); 4.54 δ (triplet, Rel.area=1.00); 7.24 δ (Rel.area=5.00).
b)
C8H10O2
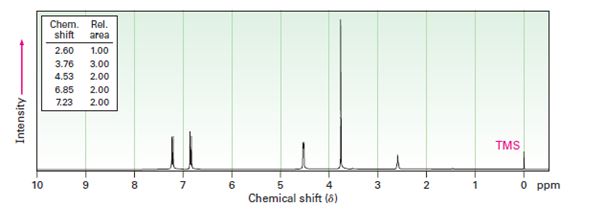
Interpretation:
A structure for the alcohol with molecular formula C8H10O2 and the following spectral data is to be proposed.
IR: 3500 cm-1,
1HNMR: 2.60 δ (Rel.area=1.00); 3.76 δ (Rel.area=3.00); 4.53 δ (Rel.area=2.00); 6.85 δ (Rel.area=2.00); 7.23 δ (Rel.area=2.00).
Concept introduction:
In 1HNMR spectrum, the alcoholic proton absorption occurs in the range 3.4 δ - 4.5 δ while that of phenolic proton occur in the range 3.0 to 8.0. The aromatic protons give a broad peak in the range 6.5 δ - 8.0 δ, the benzylic protons normally absorb in the region 2.3 δ - 3.0 δ.
To propose:
A structure for the alcohol with molecular formula C8H10O2 and the following spectral data is to be proposed.
1HNMR: 2.60 δ (Rel.area=1.00); 3.76 δ (Rel.area=3.00); 4.53 δ (Rel.area=2.00); 6.85 δ (Rel.area=2.00); 7.23 δ (Rel.area=2.00).
Trending nowThis is a popular solution!

Chapter 17 Solutions
Organic Chemistry
- Propose a structure for compound X (molecular formula C6H12O2), which gives a strong peak in its IR spectrum at 1740 cm−1. The 1H NMR spectrum of X shows only two singlets, including one at 3.5 ppm. The 13C NMR spectrum is given below. Propose a structure for X.arrow_forwardPropose a structure for the compound, C4H8O, that has the following 1H NMR spectrumarrow_forwardSynthesize the following compound from benzonitrile (C6H5CN):arrow_forward
- Treatment of (CH3)2CHCH(OH)CH2CH3 with TsOH affords two products(M and N) with molecular formula C6H12. The 1H NMR spectra of M and Nare given below. Propose structures for M and N, and draw a mechanismto explain their formation.arrow_forwardCompound P has molecular formula C5H9ClO2. Deduce the structure of P from its 1H and 13C NMR spectra.arrow_forwardCompound X (molecular formula C10H12O) was treated with NH2NH2,−OH to yield compound Y (molecular formula C10H14). Based on the 1H NMR spectra of X and Y given below, what are the structures of X and Y?arrow_forward
- Following are 1H-NMR spectra for compounds B (C6H12O2) and C (C6H10O). Upon warming in dilute acid, compound B is converted to compound C. Deduce the structural formulas for compounds B and C.arrow_forwardPropose structures for alcohols that have the following 1HNMR spectra: (a) C5H12Oarrow_forwardAldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forward
- Propose a structural formula for the analgesic phenacetin, molecular formula C10H13NO2, based on its 1H-NMR spectrum.arrow_forwardNonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardCompound H (C8H6O3) gives a precipitate when treated with hydroxylamine in aqueous ethanol and a silver mirror when treated with Tollens solution. Following is its 1H-NMR spectrum. Deduce the structure of compound H.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

