
Concept explainers
a. Which compounds are Bronsted-Lowry acids:
b. Which compounds are Bronsted-Lowry bases:
c. Classify each compound as an acid, a base, or both:
(a)
Interpretation: Among the given compounds, Bronsted-Lowry acids are to be identified.
Concept introduction: Bronsted-Lowry acids are those species which donate proton. They are also known as proton donor. The net charge on Bronsted-Lowry acids can be zero or negative or positive.
Answer to Problem 2.1P
The Bronsted-Lowry acid is
Explanation of Solution
All Bronsted-Lowry acids donate proton.
Hydrogen bromide contain hydrogen atom. Hence, it is a Bronsted-Lowry acid.
Ammonia contains a lone pair of electrons which can be easily donated to form a new bond. Hence, it is a Bronsted-Lowry base.
No hydrogen is present in the
In the given set of compounds, Bronsted-Lowry acid is
(b)
Interpretation: Among the given compounds, Bronsted-Lowry bases are to be identified.
Concept introduction: Bronsted-Lowry bases are those species which can accept a proton. The Base must contain a lone pair or pi bond that could be easily donated to an acid to form a new bond. Bronsted-Lowry base are proton acceptor. The net charge on Bronsted-Lowry base can be zero and negative.
Answer to Problem 2.1P
The Bronsted-Lowry acids are
Explanation of Solution
All Bronsted-lowry bases contain lone pair of electrons or pi bond. In
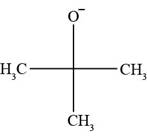
Figure 1
The molecule
![]()
Figure 2
There is neither a pi bond nor a negative charge present in
In the given set of compounds, Bronsted-Lowry acids are
(c)
Interpretation: Among the given compounds, Bronsted-lowry bases are to be identified.
Concept introduction: Bronsted-Lowry bases are those species which can accept a proton. The Base must contain a lone pair or pi bond that could be easily donated to an acid to form a new bond. Bronsted-Lowry base are proton acceptor. The net charge on Bronsted-Lowry base can be zero and negative.
Answer to Problem 2.1P
The Bronsted-Lowry acids are
Explanation of Solution
All Bronsted-Lowry bases contain lone pairs of electrons or pi bond. Bronsted-Lowry acids contain proton.
Ethanol contains hydrogen atom and lone pairs on oxygen atom. Hence, it is a Bronsted-Lowry base as well as a Bronsted-Lowry acid.
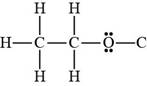
Figure 3
Propane contains only hydrogen atoms. Hence, it is a Bronsted-Lowry acid.
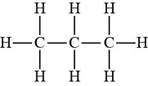
Figure 4
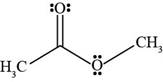
Figure 5
The Bronsted-Lowry acids are
Want to see more full solutions like this?
Chapter 2 Solutions
Organic Chemistry
- Use Table 13-3 to help answer the following questions. a. Which is the stronger base, ClO4 or C6H5NH2? b. Which is the stronger base, H2O or C6H5NH2? c. Which is the stronger base, OH or C6H5NH2? d. Which is the stronger base, C6H5NH2 or CH3NH2?arrow_forwardUse Table 14.3 to help answer the following questions. a. Which is the stronger base, ClO4 or C6H5NH2? b. Which is the stronger base, H2O or C6H5NH2? c. Which is the stronger base, OH or C6H5NH2? d. Which is the stronger base, C6H5NH2 or CH3NH2?arrow_forwardWhich is the stronger of the following two acids? (a) acetic acid, CH3CO2H, Ka = 1.8 105 (b) chloroacetic acid, ClCH2CO2H, pKa = 2.85arrow_forward
- You may need Table 13-2 to answer the following questions. a. Which is the stronger base, Cl or H2O? b. Which is the stronger base, H2O or NO2? c. Which is the stronger base, CNor OC6H5?arrow_forwardWhat is the conjugate acid of each of the following? What is the conjugate base of each?. (a) OH-. (b) H2O. (c) HCO3-. (d) NH3. (e) HSO4-. (f) H2O2. (g) HS-. (h) H5N2+arrow_forwardA hydrogen atom in the organic base pyridine, C5H5N, can be substituted by various atoms or groups to give XC5H4N, where X is an atom such as Cl or a group such as CH3. The following table gives Ka values for the conjugate acids of a variety of substituted pyridines. (a) Suppose each conjugate acid is dissolved in sufficient water to give a 0.050 M solution. Which solution would have the highest pH? The lowest pH? (b) Which of the substituted pyridines is the strongest Brnsted base? Which is the weakest Brnsted base?arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





