
Concept explainers
Rank the following ions in order of increasing basicity.
a.
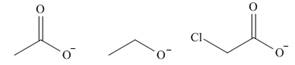
b.
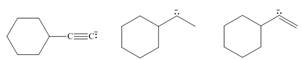
(a)
Interpretation: The order of increasing basicity for the given ions is to be ranked.
Concept introduction: A strong base possesses a weak conjugate acid and a strong acid has a weak conjugate base. In the periodic table, the basicity of ions decreases across a row as the electronegativity increases on moving from left to right and down a column as the size of atom increases on moving from top to bottom.
Answer to Problem 2.51P
The increasing order of basicity for the given ions is
Explanation of Solution
The given ions are
The increasing order of basicity for the given ions is
(b)
Interpretation: The order of increasing basicity for the given ions is to be ranked.
Concept introduction: A strong base possesses a weak conjugate acid and a strong acid has a weak conjugate base. In the periodic table, the basicity of ions decreases across a row as the electronegativity increases on moving from left to right and down a column as the size of atom increases on moving from top to bottom.
Answer to Problem 2.51P
The increasing order of basicity for the given ions is
Explanation of Solution
The given ions are
The increasing order of basicity for the given ions is
(c)
Interpretation: The order of increasing basicity for the given ions is to be ranked.
Concept introduction: A strong base possesses a weak conjugate acid and a strong acid has a weak conjugate base. In the periodic table, the basicity of ions decreases across a row as the electronegativity increases on moving from left to right and down a column as the size of atom increases on moving from top to bottom.
Answer to Problem 2.51P
The order of increasing basicity for the given ions is
Explanation of Solution
The given ions are
The conjugate acid of
The order of increasing basicity for the given ions is
(d)
Interpretation: The order of increasing basicity for the given ions is to be ranked.
Concept introduction: In hydrocarbons, alkanes, alkenes and alkynes, the order of basicity is predicted by their s-character. More is the s-character, less is the basicity or less is the s-character, more is the basicity.
Answer to Problem 2.51P
The order of increasing basicity for the given ions is shown in Figure 2.
Explanation of Solution
The given ions are,
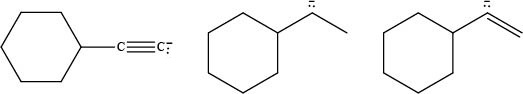
Figure 1
More is the s-character of a compound, less is the basicity. Alkanes contain
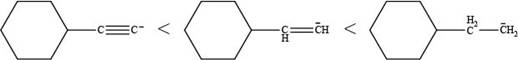
Figure 2
The order of increasing basicity for the given ions is shown in Figure 2.
Want to see more full solutions like this?
Chapter 2 Solutions
Organic Chemistry
- Consider the following four solutions: (1) apple juice, pH 3.8, (2) pickle juice, pH 3.5, (3) carbonated beverage, pH 3.0, and (4) drinking water, pH 7.2. a. Which solution has the highest [H3O+]? b. Which solution has the highest [OH]? c. List the solutions in order of increasing acidity. d. List the solutions in order of decreasing basicity.arrow_forwardUse Table 14.3 to help order the following acids from strongest to weakest HNO3,H2O,NH4+,C5H5NH+arrow_forwardWrite equations that show NH3 as both a conjugate acid and a conjugate base.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





