
(a)
Interpretation:
The complete, detailed mechanism and the major product of the given reaction are to be drawn.
Concept introduction:
Answer to Problem 20.3P
The complete mechanism of the reaction can be drawn as
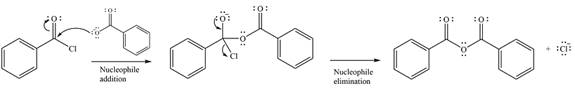
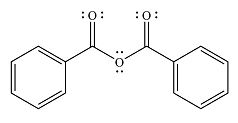
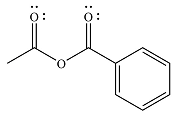
The major product of the reaction is
Explanation of Solution
The given reaction is
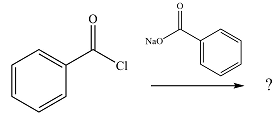
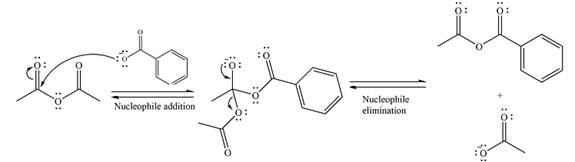
The substrate is an acid chloride, the least stable of the carboxylic acid derivatives. It will, therefore, undergo an acyl substitution via nucleophilic addition-elimination to form an acid anhydride.
The reagent is ionic, and essentially behaves as a negatively charged nucleophile, a carboxylate anion. It will attack and add to the electrophilic carbonyl carbon from the acid chloride. This will result in the formation of a tetrahedral intermediate, with the negative charge on the carbonyl oxygen of the substrate.
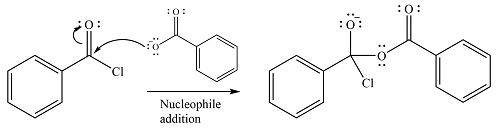
In the next step, the leaving group, chloride ion is eliminated to form the product.
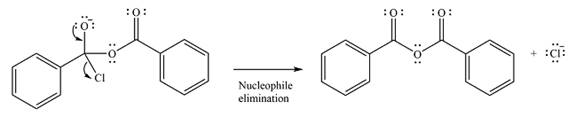
The product is more stable than the substrate, therefore, the reaction will occur.
Thus, the complete mechanism can be drawn as
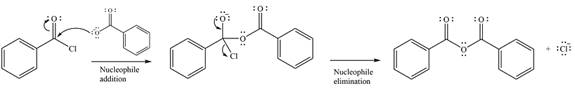
And the major product of the reaction ss

The mechanism and the major product of the given reaction were determined based on nucleophilic addition-elimination provided the possible product is of comparable or higher stability.
(b)
Interpretation:
The complete, detailed mechanism and the major product of the given reaction are to be drawn.
Concept introduction:
Carboxylic acid derivatives undergo acyl group substitution reactions when treated with appropriate nucleophiles. The reaction occurs via nucleophilic addition-elimination involving a tetrahedral intermediate. It may also involve proton transfer step(s). The reaction occurs if the possible product is more stable than the reactant. If the two are of comparable stability, the reaction will occur reversibly. The order of increasing stability of acid derivatives is
Answer to Problem 20.3P
The complete mechanism of the reaction can be drawn as
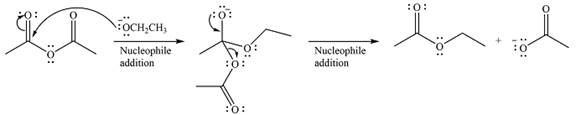
The major product of the reaction is
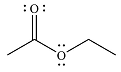
Explanation of Solution
The given reaction is
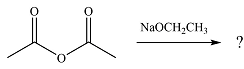
The substrate is an acid anhydride and the reagen is essentially the anionic nucleophile
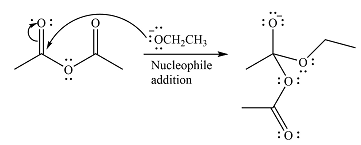
The second step is nucleophilic elimination. The acyl group from the original anhydride is eliminated to form the product, an ester.
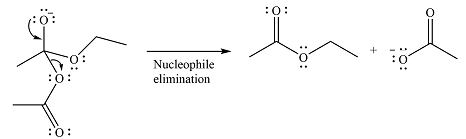
Thus, the complete mechanism can be drawn as
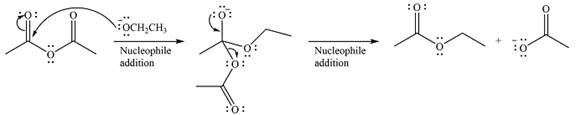
Since an ester is more stable than an acid anhydride, the reaction will occur, and the major product will be

The mechanism and the major product of the given reaction were determined based on nucleophilic addition-elimination provided the possible product is of comparable or higher stability.
(c)
Interpretation:
The complete, detailed mechanism and the major product of the given reaction are to be drawn.
Concept introduction:
Carboxylic acid derivatives undergo acyl group substitution reactions when treated with appropriate nucleophiles. The reaction occurs via nucleophilic addition-elimination involving a tetrahedral intermediate. It may also involve proton transfer step(s). The reaction occurs if the possible product is more stable than the reactant. If the two are of comparable stability, the reaction will occur reversibly. The order of increasing stability of acid derivatives is
Answer to Problem 20.3P
The complete mechanism of the reaction can be drawn as
The major product of the reaction is
Explanation of Solution
The given reaction is
The substrate is an anhydride, with electrophilic carbons. The reagent is an ionic compound, which will essentially act as an anionic nucleophile, a carboxylate ion.
In the first step, the carboxylate ion will add to one of the carbonyl carbons of the anhydride. This will result in the formation of a tetrahedral intermediate with the negaive charge shifting to the ccorresponding carbonyl oxygen.
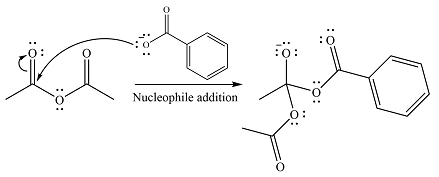
In the next step, the leaving group will be liminated as a carboxylate anion.
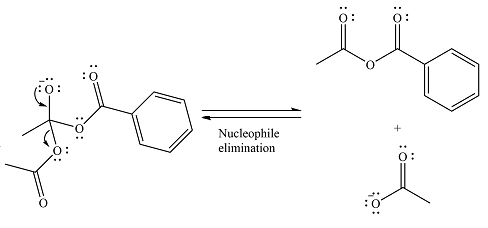
The major product is another acid anhydride of comparable stability. This means the reaction will occur, with reversible addition and elimination steps.
Thus, the complete mechanism can be drawn as
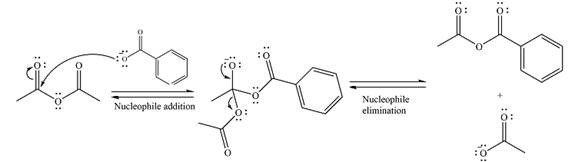
Thus, the major product will be

The mechanism and the major product of the given reaction were determined based on nucleophilic addition-elimination provided the possible product is of comparable or higher stability.
(d)
Interpretation:
The complete, detailed mechanism and the major product of the given reaction are to be drawn.
Concept introduction:
Carboxylic acid derivatives undergo acyl group substitution reactions when treated with appropriate nucleophiles. The reaction occurs via nucleophilic addition-elimination involving a tetrahedral intermediate. It may also involve proton transfer step(s). The reaction occurs if the possible product is more stable than the reactant. If the two are of comparable stability, the reaction will occur reversibly. The order of increasing stability of acid derivatives is
Answer to Problem 20.3P
There is no reaction.
Explanation of Solution
The given reaction is
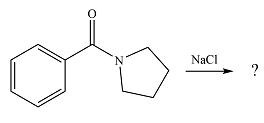
The substrate is a highly stable amide. If the
Since the possible product is of lower stability than the substrate, the reaction will not occur.
The reaction will not occur if the possible product is of lower stability than the susbtrate.
(e)
Interpretation:
The complete, detailed mechanism and the major product of the given reaction are to be drawn.
Concept introduction:
Carboxylic acid derivatives undergo acyl group substitution reactions when treated with appropriate nucleophiles. The reaction occurs via nucleophilic addition-elimination involving a tetrahedral intermediate. It may also involve proton transfer step(s). The reaction occurs if the possible product is more stable than the reactant. If the two are of comparable stability, the reaction will occur reversibly. The order of increasing stability of acid derivatives is
Answer to Problem 20.3P
There is no reaction.
Explanation of Solution
The given reaction is
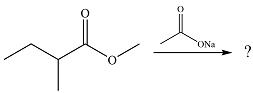
The substrate is an ester while the possible product of the reaction would be an acid anhydride. Since an ester is more stable than an acid anhydride, the reaction will not occur.
The reaction will not occur if the possible product is of lower stability than the susbtrate.
(f)
Interpretation:
The complete, detailed mechanism and the major product of the given reaction are to be drawn.
Concept introduction:
Carboxylic acid derivatives undergo acyl group substitution reactions when treated with appropriate nucleophiles. The reaction occurs via nucleophilic addition-elimination involving a tetrahedral intermediate. It may also involve proton transfer step(s). The reaction occurs if the possible product is more stable than the reactant. If the two are of comparable stability, the reaction will occur reversibly. The order of increasing stability of acid derivatives is
Answer to Problem 20.3P
The complete mechanism of the reaction can be drawn as
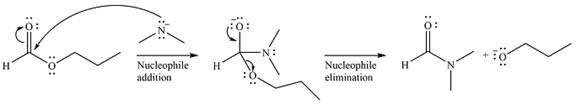
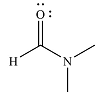
The major product of the reaction is
Explanation of Solution
The given reaction is
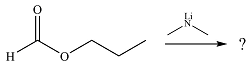
The substrate is an ester, with an electrophilic carbonyl carbon. The reagent is essentially an anionic nucleophile
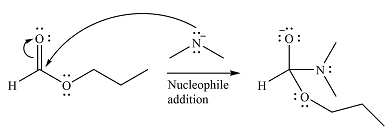
In the second step, the alkoxide group from the original ester will be eliminated to form the product, an amide.
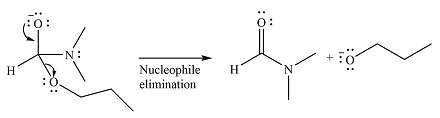
Since the product is more stable than the substrate, the reaction will occur.
Thus, the complete mechanism can be drawn as

And the product of the reaction will b

The mechanism and the major product of the given reaction were determined based on nucleophilic addition-elimination provided the possible product is of comparable or higher stability.
Want to see more full solutions like this?
Chapter 20 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Draw the complete, detailed mechanism for each of the following reactions and predict the major products.arrow_forwardDraw the complete, detailed mechanism for the reaction shown here and give the major product. CH3I (excess) ? NH2arrow_forwardThe reaction shown here is an example of the Favorskii reaction, which involves an R¯ leaving group in a nucleophilic addition-elimination reaction. (a) Draw the complete, detailed mechanism for this reaction and explain why R can act as a leaving NaOH H2O group. (b) Suggest how you can synthesize an ester from cyclopropanone using only this reaction.arrow_forward
- please help with this question thank you. Draw the mechanism for the reaction given.arrow_forwardOH Draw the complete, detailed mechanism for the reaction shown here. (See Problem 17.63.) NaBH4 Ethanolarrow_forwardDraw a complete mechanism and predict the major product in the following reactionarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





