
Concept explainers
(a)
Interpretation:
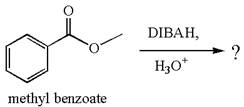
The product with detailed mechanism for the reaction between methyl benzoate and
Concept introduction:
An ester can be converted to a primary alcohol by reacting with
Answer to Problem 20.46P
The product with detailed mechanism for the reaction between methyl benzoate and
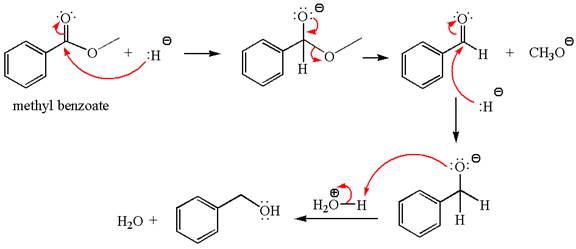
Explanation of Solution
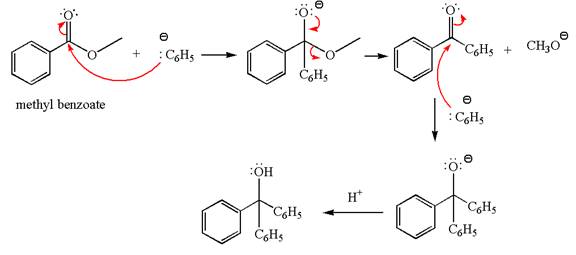
The equation for the reaction of methyl benzoate with
Methyl benzoate is an ester, which, on reaction with
The product with detailed mechanism for the given reaction is drawn based on the reactivity of
(b)
Interpretation:
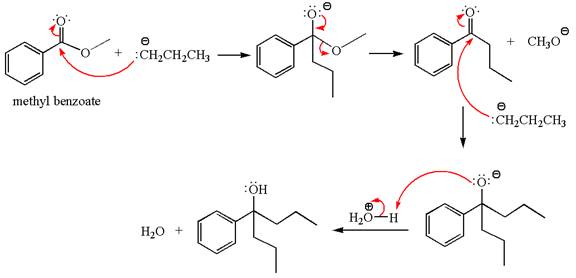
The product with detailed mechanism for the reaction between methyl benzoate and
Concept introduction:
An ester can be converted to a tertiary alcohol by reacting it with
Answer to Problem 20.46P
The product with detailed mechanism for the reaction between methyl benzoate and
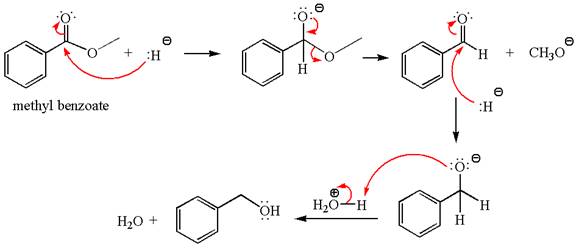
Explanation of Solution
The equation for the reaction of methyl benzoate with
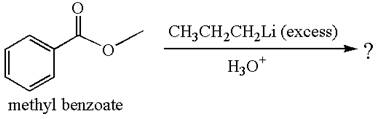
Methyl benzoate is an ester, which, on reaction with
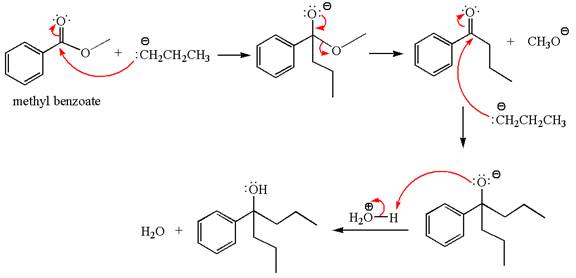
The product with detailed mechanism for the given reaction is drawn based on the reactivity of
(c)
Interpretation:
Whether methyl benzoate can react with
Concept introduction:
The reagent
Answer to Problem 20.46P
Mmethyl benzoate cannot react with
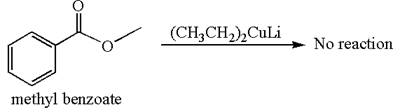
Explanation of Solution
The equation for the reaction of methyl benzoate with
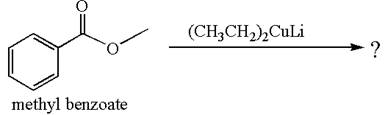
Methyl benzoate is an ester and
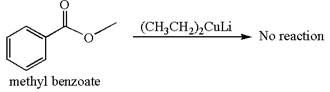
It is determined that no reaction occurs based on the reactivity of
(d)
Interpretation:
The product with detailed mechanism for the reaction between methyl benzoate and
Concept introduction:
An ester can be converted to a tertiary alcohol by reacting it with
Answer to Problem 20.46P
The product with detailed mechanism for the reaction between methyl benzoate and
Explanation of Solution
The equation for the reaction of methyl benzoate with
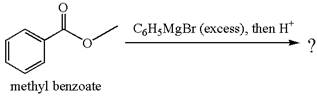
Methyl benzoate is an ester, which, on reaction with

The product with detailed mechanism for the given reaction is drawn based on the reactivity of
(e)
Interpretation:
The product with detailed mechanism for the reaction between methyl benzoate and
Concept introduction:
An ester can be reduced to aldehyde without reducing it further to alcohol using a specific reagent such as
Answer to Problem 20.46P
The product with detailed mechanism for the reaction between methyl benzoate and
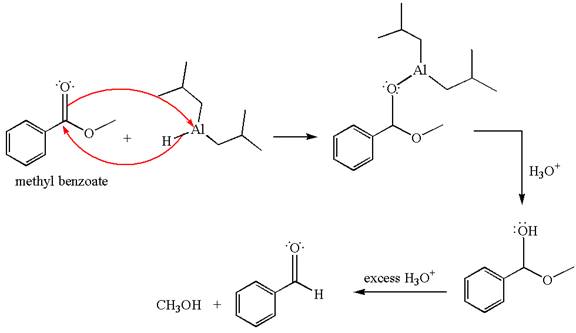
Explanation of Solution
The equation for the reaction of methyl benzoate with
Methyl benzoate is an ester, which, on reaction with
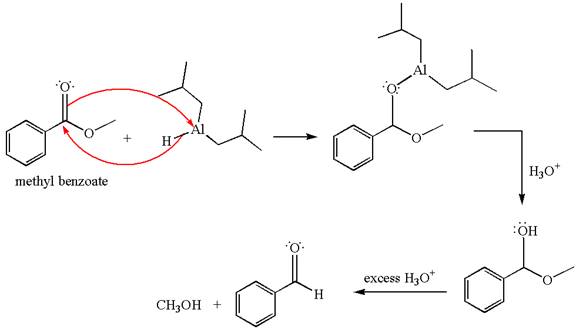
The product with detailed mechanism for the given reaction is drawn based on the reactivity of
Want to see more full solutions like this?
Chapter 20 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Determine whether each of the following syntheses requires a reaction that alters the carbon skeleton. (a) (b) OH (c) (d) OCH,CH3 CH3 (e) OH (f) H,CO он H;CO. (g) H3CO, (h) H3CO. NO2 NO2arrow_forwarda) Provide a mechanism that accounts for the product formed in the following reaction. OH CH H" ► 1-ethoxy-1-methyleyclohexane + CH,CH,OH b) Show the structure of the product(s) formed in the following reaction and write the mechanism for the reaction. OH CH + (CH;);COHarrow_forwardDraw the structure of each of the following molecules. (a) pentanoyl chloride; (b) 4-(2-methylpropyl)heptanedioyl chloride;(c) (S)-5-phenyloctanoyl chloridearrow_forward
- 4. identify the structure of A and B in the following synthetic scheme: Write out complete reactions for each step, showing the structure of all reactants and products. (a) cyclohexanol +Na2Cr2O7/H2SO4, H2O -------> A (b) A + Et2NH/H2SO4 ----->Barrow_forward4) Propose a synthetic pathway to prepare 2-methyl-propan-2-ol from propane. You're allowed to use any reagents you like but all the carbon atoms in your product should come from the propane. он It must be propanone otherwise the conversion would be too long.arrow_forwardChlorination of 2-butanone yields two isomeric products, each having the molecular formula C4H7ClO. (a) What are these two compounds? (b) Write structural formulas for the enol intermediates that lead to each of these compounds. (c) Using curved arrows, show the flow of electrons in the reaction of each of the enols with Cl2.arrow_forward
- Predict the intermediates and final product. Provide a rational mechanism.arrow_forward(a) In an acid-catalyzed hydration, one of the following 10 carbon alkynes is expected to produce a single hydration product? Select the correct alkyne and draw the structure of the hydration product that is formed from this alkyne. (I) 2-decyne; (II) 3-decyne; (III) 4-decyne; (IV) 5-decyne; (V) none of these will give a single hydration product. (b) The reaction shown below gives Compound X as the major product. The mass spectrum of X is shown. Br2, H20 Compound X 100 - MS-IW-5644 80 60 40 - 20 - 20 40 60 80 100 120 140 160 180 200 220 m/z Considering the reactions of alkynes and the MS data, de duce which of following structures corresponds to X: Br Br HO, IV V I II II Support your answer with a reaction mechanism for fomation of X and identification of relevant peaks in the mass spectrum. 12 Relative Intensityarrow_forward(c)Show step by step how to synthesize methoxybenzene from benzene.arrow_forward
- An important step in one synthesis of carboxylic acids is the deprotonation of diethyl malonate and its alkyl-substituted derivative: Base CH;CH2O OCH,CH3 CH;CH,0 OCH2CH3 H2 Diethyl malonate Base CH;CH,0 °C `OCH,CH3 CH;CH,O OCH,CH3 R Alkyl substituted diethyl malonate NaOH can deprotonate diethyl malonate effectively, but NaOC(CH3)3 is typically used to deprotonate the alkyl-substituted derivative. Explain why.arrow_forward3) Beginning from acetylene and any alkyl halide needed, how would you prepare the following compound, there may be more than one step. HC=CH CH;CH,CH,CH,CH,CH,CH;arrow_forwardDraw the structure of each of the following molecules (a) 2,2-dimethylcyclopentane-1-carboxylic acid;(b) (R)-3-chloropentanoic acid; (c) (2R,3S)-2,3-dinitrobutanedioic acidarrow_forward
