
Concept explainers
(a)
Interpretation:
Whether the given compound is synthesized in good yield or not by an aldol condensation with its starting materials is to be stated. The reason as to why the required aldol condensation would not succeed for compound that is not synthesized in good yield is to be stated.
Concept Introduction:
Aldol condensation is the nucleophilic addition reaction of the carbonyl compounds. An enolate ion generated from one of the carbonyl compound attack the electrophilic carbon of the other carbonyl compound to produce
Answer to Problem 22.22P
The given compound can be synthesized using aldol condensation in good yield. The starting materials are
Explanation of Solution
The given compound is shown below.
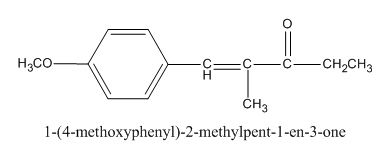
Figure 1
In the above preparation of the compound, the enolate is generated on the
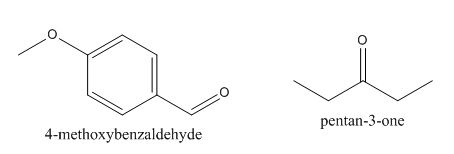
Figure 2
The given compound can be synthesized by aldol condensation of
(b)
Interpretation:
Whether the given compound is synthesized in good yield or not by an aldol condensation with its starting materials is to be stated. The reason as to why the required aldol condensation would not succeed for compound that is not synthesized in good yield is to be stated.
Concept Introduction:
Aldol condensation is the nucleophilic addition reaction of the carbonyl compounds. An enolate ion generated from one of the carbonyl compound attack the electrophilic carbon of the other carbonyl compound to produce
Answer to Problem 22.22P
The given compound can be synthesized using aldol condensation in good yield. The starting materials are benzaldehyde and
Explanation of Solution
The given compound is shown below.
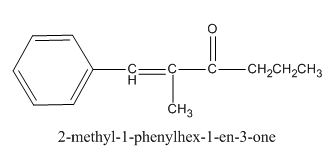
Figure 3
In the preparation of the above compound, the enolate will be generated on the
The structures of the starting materials which are used to produce the given compound are shown below.
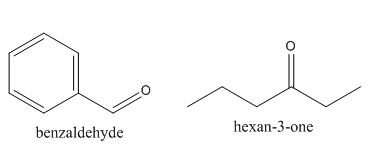
Figure 4
The given compound can be synthesized by aldol condensation of benzaldehyde and
(c)
Interpretation:
Whether the given compound is synthesized in good yield or not by an aldol condensation with its starting materials is to be stated. The reason as to why the required aldol condensation would not succeed for compound that is not synthesized in good yield is to be stated.
Concept Introduction:
Aldol condensation is the nucleophilic addition reaction of the carbonyl compounds. An enolate ion generated from one of the carbonyl compound attack the electrophilic carbon of the other carbonyl compound to produce
Answer to Problem 22.22P
The given compound can be synthesized using aldol condensation but not in a good in good yield. The starting material is
Explanation of Solution
The given compound is shown below.
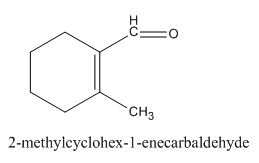
Figure 5
In the preparation of the above compound, intramolecular aldol condensation takes place. To form this compound enolate must be generated by aldehyde and it has to attack the carbonyl carbon of ketone which is not favorable condition. Favorable condition is that enolate should be generated on ketone which attacks the electrophilic carbon of the aldehyde. This is because the carbon of aldehyde is more acidic than the carbon of ketone. The structure of the starting material which is used to produce the given compound is shown below.
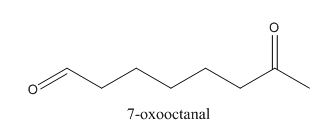
Figure 6
Therefore, the given compound cannot be synthesized by aldol condensation in a good yield.
This compound cannot be synthesized in a good yield by the aldol condensation.
(d)
Interpretation:
Whether the given compound is synthesized in good yield or not by an aldol condensation with its starting materials is to be stated. The reason as to why the required aldol condensation would not succeed for compound that is not synthesized in good yield is to be stated.
Concept Introduction:
Aldol condensation is the nucleophilic addition reaction of the carbonyl compounds. An enolate ion generated from one of the carbonyl compound attack the electrophilic carbon of the other carbonyl compound to produce
Answer to Problem 22.22P
The given compound can be synthesized by aldol condensation in good yield. The starting material is
Explanation of Solution
The given compound is shown below.
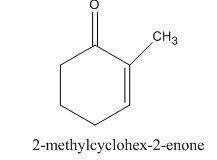
Figure 7
The given compound, is prepared by the intramolecular aldol condensation of
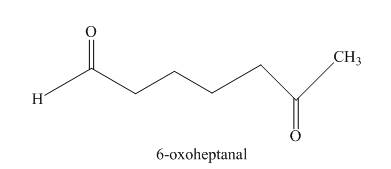
Figure 8
The given compound can be synthesized by intramolecular aldol condensation of
(e)
Interpretation:
Whether the given compound is synthesized in good yield or not by an aldol condensation with its starting materials is to be stated. The reason as to why the required aldol condensation would not succeed for compound that is not synthesized in good yield is to be stated.
Concept Introduction:
Aldol condensation is the nucleophilic addition reaction of the carbonyl compounds. An enolate ion generated from one of the carbonyl compound attack the electrophilic carbon of the other carbonyl compound to produce
Answer to Problem 22.22P
The given compound can be synthesized from aldol condensation in a good yield. The starting materials are:
Explanation of Solution
The given compound is shown below.
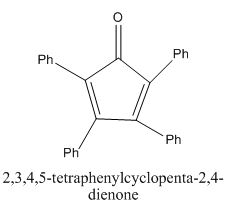
Figure 9
In the preparation of the given compound, the enolate will be generated on the
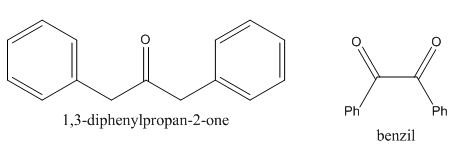
Figure 10
The given compound can be synthesized by aldol condensation of
(f)
Interpretation:
Whether the given compound is synthesized in good yield or not by an aldol condensation with its starting materials is to be stated. The reason as to why the required aldol condensation would not succeed for compound that is not synthesized in good yield is to be stated.
Concept Introduction:
Aldol condensation is the nucleophilic addition reaction of the carbonyl compounds. An enolate ion generated from one of the carbonyl compound attack the electrophilic carbon of the other carbonyl compound to produce
Answer to Problem 22.22P
The given compound can be synthesized by aldol condensation in a good yield. The starting materials are
Explanation of Solution
The given compound is shown below.
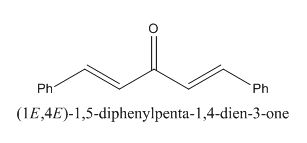
Figure 11
In the preparation of the compound, the enolate is generated on the
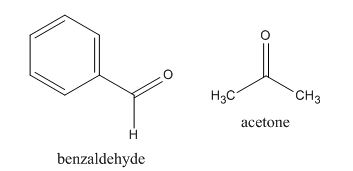
Figure 12
The given compound can be synthesized by aldol condensation of benzaldehyde and acetone.
(g)
Interpretation:
Whether the given compound is synthesized in good yield or not by an aldol condensation with its starting materials is to be stated. The reason as to why the required aldol condensation would not succeed for compound that is not synthesized in good yield is to be stated.
Concept Introduction:
Aldol condensation is the nucleophilic addition reaction of the carbonyl compounds. An enolate ion generated from one of the carbonyl compound attack the electrophilic carbon of the other carbonyl compound to produce
Answer to Problem 22.22P
The given compound can be synthesized by aldol condensation. The starting materials are trimethylacetone and acetaldehyde.
Explanation of Solution
The given compound is shown below.
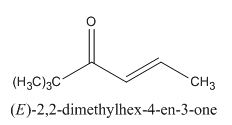
Figure 13
In the preparation of the given compound, the enolate will be generated on the
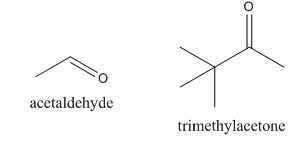
Figure 14
The given compound can be synthesized by aldol condensation by trimethylacetone and acetaldehyde in a good yield.
(h)
Interpretation:
Whether the given compound is synthesized in good yield or not by an aldol condensation with its starting materials is to be stated. The reason as to why the required aldol condensation would not succeed for compound that is not synthesized in good yield is to be stated.
Concept Introduction:
Aldol condensation is the nucleophilic addition reaction of the carbonyl compounds. An enolate ion generated from one of the carbonyl compound attack the electrophilic carbon of the other carbonyl compound to produce
Answer to Problem 22.22P
The given compound can be synthesized by aldol condensation in a good yield. The starting material is
Explanation of Solution
The given compound is shown below.
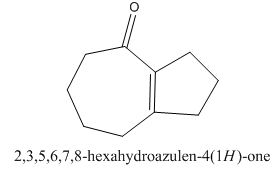
Figure 15
The enolate will be generated on the
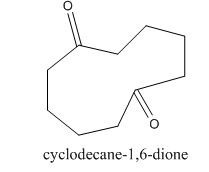
Figure 16
The given compound can be synthesized by aldol condensation of
Want to see more full solutions like this?
Chapter 22 Solutions
Organic Chemistry
- Provide a chemical reaction on how the following compounds can be synthesized via aldol condensation. note that the starting reactants should be benzaldehyde and a ketonearrow_forwardShow how the following ketones might be synthesized from the indicated acids, usingany necessary reagents.(b) methyl cyclohexyl ketone from cyclohexanecarboxylic acidarrow_forwardExplain how benzaldehyde and dimedone reacts with each other, and then with the aminotriazole to form compound 1a in the presence of an acid catalyst. Provide a detailed reaction mechanism and explanation.arrow_forward
- Acid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forwardThe following questions pertain to the esters shown and behavior under conditions of the Claisen condensation.(a) Two of these esters are converted to β-keto esters in good yield on treatment with sodium ethoxide and subsequent acidification of the reaction mixture. Which two are these? Write the structure of the Claisen condensation product of each one. (b) One ester is capable of being converted to a β-keto ester on treatment with sodium ethoxide, but the amount of β-keto ester that can be isolated after acidification of the reaction mixture is quite small. Which ester is this? (c) One ester is incapable of reaction under conditions of the Claisen condensation. Which one? Why?arrow_forward• Using aldol or crossed aldol condensation, suggest a synthesis of the following compounds: a b مليمت H3C H3C CH3 H3C ge H Savedarrow_forward
- Compound A undergoes an acid-catalyzed hydrolysis. One of the products (B) that is isolated gives the following 1H NMR spectrum. Identify the compounds A and Carrow_forwardWhat is the structural requirement of the following reactions? 1) Iodoform Reaction 2) Aldol Condensationarrow_forwardPredict the products when each compound undergo Ozonolysis (a) (b) (c) , (d) (e)arrow_forward
- The following intermediate was needed for the synthesis of tamoxifen, a widely used antiestrogen drug for treating estrogen-dependent cancers such as breast and ovarian cancer. Needed for the (A) synthesis of tamoxifen Propose a synthesis for this intermediate from compound A.arrow_forwardThe following compound may be synthesized through alkylation of an appropriate enamine with an alkyl bromide, followed by hydrolysis of the resulting immonium ion. Using this strategy, provide the necessary starting materials for the synthesis. Draw the necessary enamine below.arrow_forwardThe Stork reaction is a condensation reaction between an enamine donor and an α,β-unsaturated carbonyl acceptor. The overall reaction consists of a three-step sequence of formation of an enamine from a ketone, Michael addition to an α,β-unsaturated carbonyl compound, and hydrolysis of the enamine in dilute acid to regenerate the ketone. Consider the Stork reaction between acetophenone and 3-buten-2-one. Draw the structure of the product of the enamine formed between acetophenone and pyrrolidine. Draw the structure of the Michael addition product. Draw the structure of the final product.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
