
Concept explainers
(a)
Interpretation:
The synthesis of
Concept introduction:
In
Answer to Problem 22.82AP
The synthesis of
Explanation of Solution
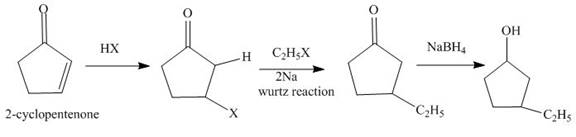
The reaction of
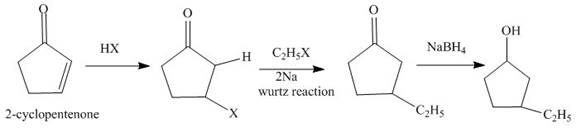
Figure 1
The synthesis of
(b)
Interpretation:
The synthesis of
Concept introduction:
In organic chemistry, the class of functional group which is derived from conjugated diene by replacing one double bond with a carbonyl group. The dienone compound is in conjugation to each other.
Answer to Problem 22.82AP
The synthesis of
Explanation of Solution
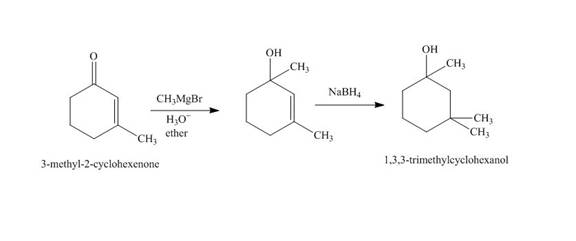
The reaction of
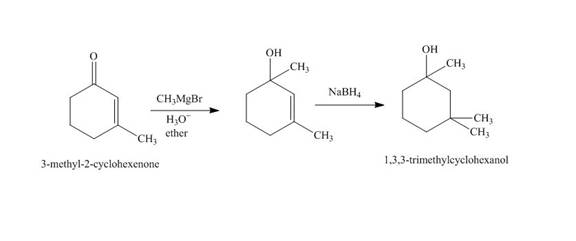
Figure 2
The synthesis of
(c)
Interpretation:
The synthesis of
Concept introduction:
An ester is a derivative of carboxylic which is obtained by replacing the
Answer to Problem 22.82AP
The synthesis of
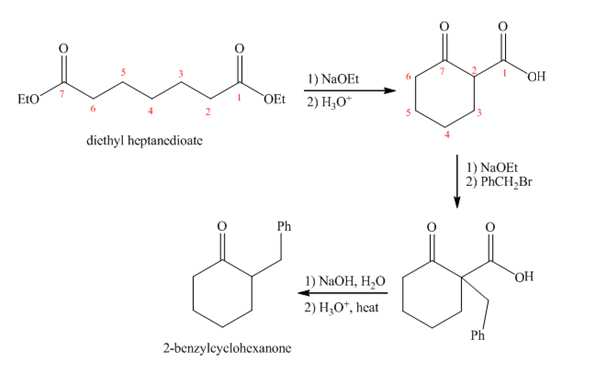
Explanation of Solution
Diethyl heptanedioate in presence of

Figure 3
The synthesis of
(d)
Interpretation:
The synthesis of
Concept introduction:
An ester is a derivative of carboxylic which is obtained by replacing the
Answer to Problem 22.82AP
The synthesis of
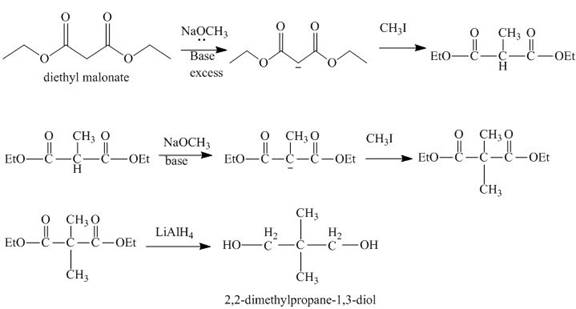
Explanation of Solution
The reaction of diethyl malonate in presence of base and then

Figure 4
The synthesis of
(e)
Interpretation:
The synthesis of
Concept introduction:
An ester is a derivative of carboxylic acid which is obtained by replacing the
Answer to Problem 22.82AP
The synthesis of
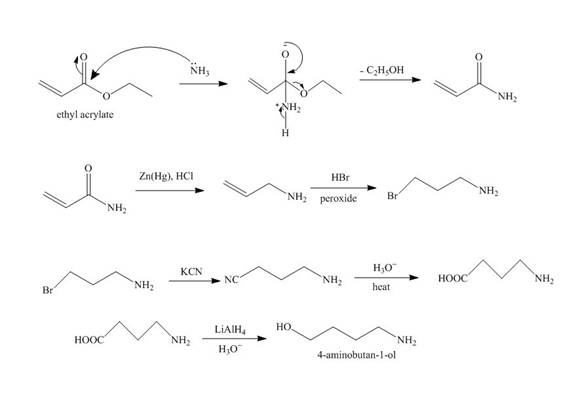
Explanation of Solution
The reaction of ethyl acrylate with ammonia forms amide derivative. It undergoes reduction reaction with

Figure 5
The synthesis of
(f)
Interpretation:
The synthesis of
Concept introduction:
In organic chemistry, the compound with the molecular formula
Answer to Problem 22.82AP
The synthesis of
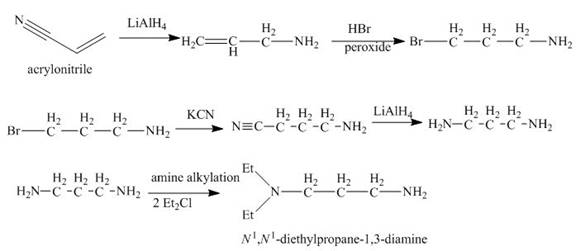
Explanation of Solution
The reaction of acrylonitrile with
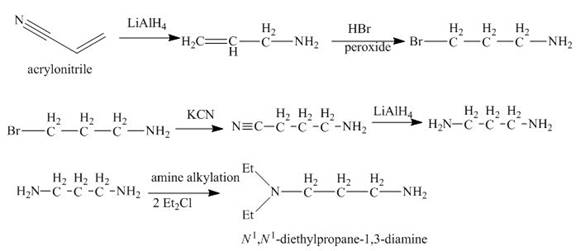
Figure 6
The synthesis of
(g)
Interpretation:
The synthesis of
Concept introduction:
Carboxylic acid is a class of organic compound contains a
Answer to Problem 22.82AP
The synthesis of
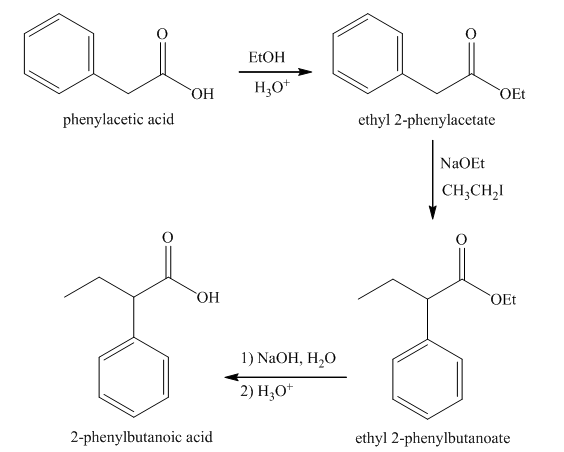
Explanation of Solution
Esterification of phenylacetic acid forms ethyl

Figure 7
The synthesis of
(h)
Interpretation:
The synthesis of
Concept introduction:
In Aldol condensation reaction, the base abstracts an acidic proton from the
Answer to Problem 22.82AP
The synthesis of
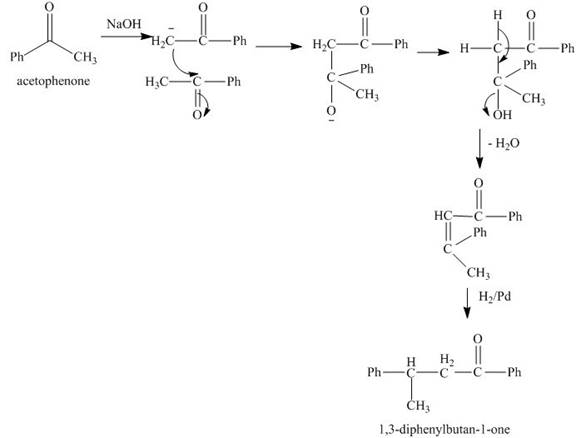
Explanation of Solution
The reaction of acetophenone in presence of base abstracts a

Figure 8
The synthesis of
(i)
Interpretation:
The synthesis of deuterium substituted diphenylethanol from phenylacetic acid is to be stated.
Concept introduction:
Carboxylic acid is a class of organic compound that contains a
Answer to Problem 22.82AP
The synthesis of di-deuterium substituted diphenyl ethanol from phenylacetic acid is shown below.
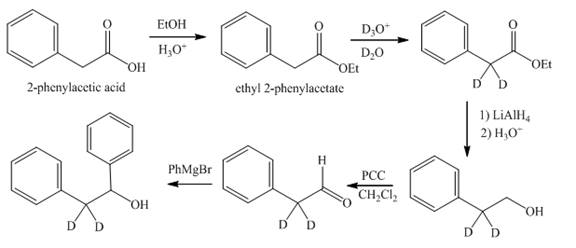
Explanation of Solution
Reaction of

Figure 9
The synthesis of deuterium substituted diphenyl ethanol from phenylacetic acid is shown in Figure 9.
(j)
Interpretation:
The synthesis of tetra-deuterium substituted product from cyclohexanone is to be stated.
Concept introduction:
In organic chemistry, the carbonyl compound is a class of functional group which contains a
Answer to Problem 22.82AP
The synthesis of tetra-deuterium substituted product from cyclohexanone is shown below.
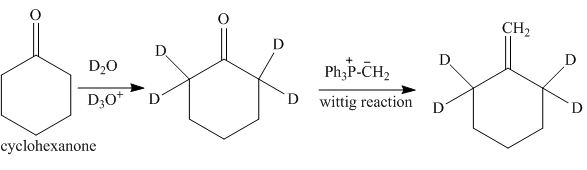
Explanation of Solution
The reaction of cyclohexanone with

Figure 10
The synthesis of tetra-deuterium substituted from cyclohexanone is shown in Figure 10.
(k)
Interpretation:
The synthesis of
Concept introduction:
Friedel Craft acylation is an electrophilic
Answer to Problem 22.82AP
The synthesis of
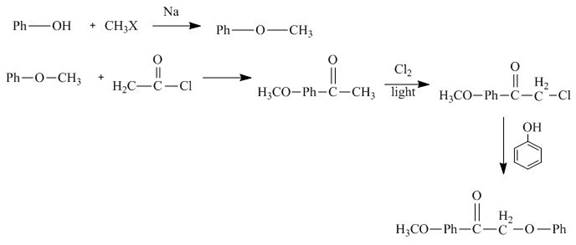
Explanation of Solution
The reaction of phenol with methyl halide forms methylphenyl ether. It reacts with acetyl chloride followed by chlorination in sunlight. Then it reacts with phenol to give the desired product. The synthesis of

Figure 11
The synthesis of
(l)
Interpretation:
The synthesis of
Concept introduction:
An ester is a derivative of carboxylic which is obtained by replacing the
Answer to Problem 22.82AP
The synthesis of
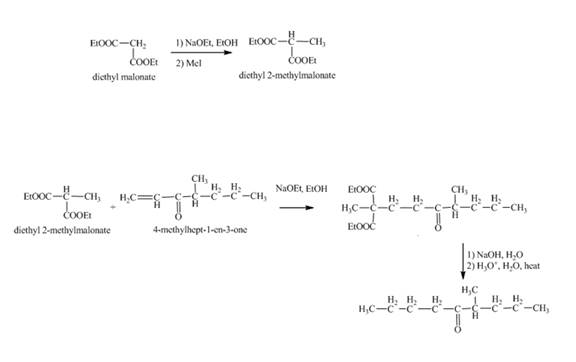
Explanation of Solution
Methylation of diethyl malonate in the presence of sodium ethoxide and

Figure 12
The synthesis of
(m)
Interpretation:
The synthesis of
Concept introduction:
An ester is a derivative of carboxylic which is obtained by replacing the
Answer to Problem 22.82AP
The synthesis of
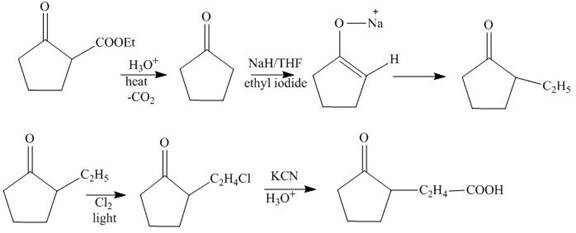
Explanation of Solution
The hydrolysis reaction of

Figure13
The synthesis of
Want to see more full solutions like this?
Chapter 22 Solutions
Organic Chemistry
- Acid-catalyzed dehydration of 3-methyl-2-butanol gives three alkenes: 2-methyl-2- butene, 3-methyl-1-butene, and 2-methyl-1-butene. Propose a mechanism to account for the formation of each productarrow_forwardPrepare the following compounds : 1-Starting with benzene or any other reagents , prepare 3-phenyl -1-propanolarrow_forwardElectrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict the major products of the reactions of naphthalene with HNO3, H2SO4.arrow_forward
- Provide reagents/conditions to accomplish the following syntheses. Several steps are required in some cases. H2N NH2 Но TH. NH2 HOarrow_forward1. At what position and on what ring would you expect the following substances to undergo electrophilic substitution? (b) CH3 Br lel CH3 2. Rank the compounds in each group according to their reactivity toward electrophilic substitution. (a) Chlorobenzene, o-dichlorobenzene, benzene (b) p-Bromonitrobenzene, nitrobenzene, phenol (c) Fluorobenzene, benzaldehyde, 0-xylene (d) Benzonitrile, p-methylbenzonitrile, p-methoxybenzonitrilearrow_forwardAldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forward
- Choose reagents to convert 2-cyclohexenone to the following compounds.arrow_forwardFollowing are 1H-NMR spectra for compounds B (C6H12O2) and C (C6H10O). Upon warming in dilute acid, compound B is converted to compound C. Deduce the structural formulas for compounds B and C.arrow_forwardComplete the following transformations by providing reagents or products:arrow_forward
