
Concept explainers
A mass of 3 kg of saturated liquid–vapor mixture of water is contained in a piston–cylinder device at 160 kPa. Initially, 1 kg of the water is in the liquid phase and the rest is in the vapor phase. Heat is now transferred to the water, and the piston, which is resting on a set of stops, starts moving when the pressure inside reaches 500 kPa. Heat transfer continues until the total volume increases by 20 percent. Determine (a) the initial and final temperatures, (b) the mass of liquid water when the piston first starts moving, and (c) the work done during this process. Also, show the process on a P-v diagram.
FIGURE P4–120
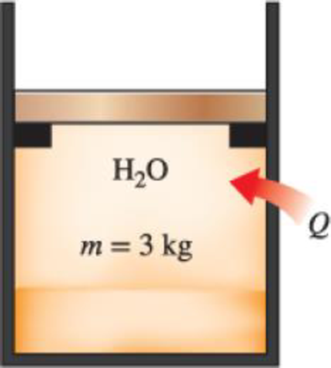
(a)
The initial temperature of the piston cylinder device.
The final temperature of the piston cylinder device.
Answer to Problem 120RP
The initial temperature of the piston cylinder device is
The final temperature of the piston cylinder device is
Explanation of Solution
Determine the total initial volume of piston cylinder device.
Here, the mass of the liquid phase is
Determine the total volume of the piston cylinder device at final state.
Determine the specific volume of the piston cylinder device at final state.
Here, the mass of the saturated liquid vapour mixture of water is contained in a piston cylinder device is
Conclusion:
Write the formula of interpolation method of two variables.
Here, the variables denote by x and y is saturated pressure and saturated temperature.
For initial temperature of the piston cylinder device.
Show the temperature at pressure of 150 kPa, 160 kPa, and 175 kPa as in Table (1).
|
Pressure, kPa |
Temperature, C |
| 150 kPa | 111.35 |
| 160 kPa | |
| 175 kPa | 116.04 |
Substitute the value of x and y from Table (1) in Equation (IV) to calculate the value of initial temperature
Thus, the initial temperature of the piston cylinder device is
For specific volume of saturated liquid of the piston cylinder device.
Show the specific volume of saturated liquid at pressure of 150 kPa, 160 kPa, and 175 kPa as in Table (2).
|
Pressure, kPa |
Specific volume of saturated liquid, |
| 150 kPa | 0.001053 |
| 160 kPa | |
| 175 kPa | 0.001057 |
Substitute the value of x and y from Table (2) in Equation (IV) to calculate the value of specific volume of saturated liquid
For specific volume of saturated vapour of the piston cylinder device.
Show the specific volume of saturated vapour at pressure of 150 kPa, 160 kPa, and 175 kPa as in Table (3).
|
Pressure, kPa |
Specific volume of saturated vapour, |
| 150 kPa | 1.1594 |
| 160 kPa | |
| 175 kPa | 1.0037 |
Substitute the value of x and y from Table (3) in Equation (IV) to calculate the value of specific volume of saturated vapour
Substitute
Substitute
Substitute
The unit conversion of pressure from kPa to MPa.
For temperature of the piston cylinder device at final state.
Show the temperature at specific volume of the piston cylinder device at final state at
|
specific volume of the piston cylinder device at final state, |
Temperature, |
| 600 | |
| 700 |
Substitute the value of x and y from Table (4) in Equation (IV) to calculate the value of temperature of the piston cylinder device at final state
Thus, the final temperature of the piston cylinder device is
(b)
The mass of liquid water when the piston first starts moving.
Answer to Problem 120RP
The mass of liquid water when the piston first starts moving is
Explanation of Solution
Determine the specific volume of the piston cylinder device at this state.
Here, the mass of the saturated liquid vapour mixture of water is contained in a piston cylinder device is
Conclusion:
Since,
Substitute
Therefore, the value of specific volume of the piston cylinder device at this state is greater than
Thus, the mass of liquid water when the piston first starts moving is
(c)
The work done during the process state 2 and 3.
Answer to Problem 120RP
The work done during the process state 2 and 3 is
Explanation of Solution
Determine the work done in constant pressure process.
Conclusion:
Substitute
Thus, the work done during the process state 2 and 3 is
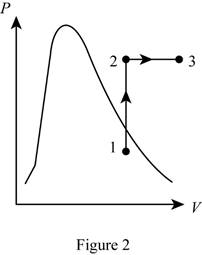
Show the P-v diagram of this process.
Want to see more full solutions like this?
Chapter 4 Solutions
Thermodynamics: An Engineering Approach
- Initially, a rigid tank contains 500L of water at 300°C. By volume, the tank contains 60% liquid and 40% vapor. At the bottom of the tank, as shown, a valve is opened and half of the mass of water is withdrawn which is in the liquid phase. Heat transfer takes place such that the temperature within the tank is constant. Determine the heat transfer in kJ required to withdraw half of its mass and maintain its temperature.arrow_forwardA piston–cylinder device contains 0.05 m3 of a gas initially at 200 kPa. At this state, a linear spring that has a spring constant of 150 kN/m is touching the piston but exerting no force on it. Now heat is transferred to the gas, causing the piston to rise and to compress the spring until the volume inside the cylinder doubles. If the cross-sectional area of the piston is 0.25 m2, determine (a) the final pressure inside the cylinder, (b) the total work done by the gas, and (c) the fraction of this work done against the spring to compress it.arrow_forwardInitially, a rigid tank contains 500L of water at 300°C. By volume, the tank contains60% liquid and 40% vapor. At the bottom of the tank, as shown, a valve is opened and half of the mass of water is withdrawn which is in the liquid phase. Heat transfer takes place such that the temperature within the tank is constant. Determine the heat transfer in kJ required to withdraw half of its mass and maintain its temperature.arrow_forward
- 2. A rigid vessel contains 5kg of wet steam (water) at 0.4 MPa. After the addition of 9585 kJ of heat the steam has a pressure of 2.0 MPa and a Temperature of 7000C Determine (a.) the initial quality, (b.) initial internal energy and (c.) initial specific volume of the steam.arrow_forwardA piston-cylinder device with a set of stops initially contains 0.3 kg of steam at 1.0 MPa and 400 °C. The location of the stops corresponds to 60% of the initial volume. Now, steam loses heat to the surroundings and the piston moves down hitting the set of stops. After the piston hits the set of stops, the cooling continues until the cylinder contains water at 100 °C. Determine: (a) the initial volume of steam (b) the total heat transfer (Q) of the steam by the time the piston first hits the stops (c) the final pressure and the quality (if mixture) when the water cools to 100 °C.arrow_forwardA piston-cylinder device initially contains 0.4 m^3 of saturated water vapor at a pressure of 200 kPa. At this time, a linear spring (K = 150 N/m, no other information about spring provided) is touching the piston, but exerts no force upon it. Heat is now transferred to the water vapor for 30 minutes through a resistive heater having a current of 10 amperes. The pressure and volume rise to 300 kPa and 0.6 m^3 while the system lost 10 kJ of heat during this process. Determine the voltage of the heater. Show detailed calculations and specify any required values found on thermodynamic tables.arrow_forward
- Water is heated in a piston/cylinder arrangement at constant pressure. Initially the water is saturated vapor at 200 kPa and has a specific volume of 0.88578 m/kg. At the final state the temperature of the water is 300 C and has a specific volume of 1.31623 m/kg. The mass of the water is 2 kg. In the question that follows, select the answer that is closest to the true value. What is the work done by the water in units of kJ ?arrow_forwardInitially saturated water liquid at 200°C is contained in a piston-cylinder. The water is then heated isothermally until its volume is 100 times larger than its initial volume. 1. Determine the increase in energy (kJ/kg) of the water. 2. Determine the work transfer (kJ/kg) and indicate whether it is into or out of the system. 3. Determine the heat transfer (kJ/kg) and indicate whether it is into or out of the system. saturated water 2000 Earrow_forward2. A well-insulated piston-cylinder device contains 5 L of saturated water at a constant pressure of 150 kPa. Water is stirred by a propeller connected to a 500 W motor while a power source that supplied 8A of electrical current flow through the coil. The propeller and coil are placed in the water for which they are switched ON for 38 minutes. If one-half of the mass of liquid is vaporized to saturation in this process, determine (a) is this nonflow or steady flow process? (b) draw the pv and Ts diagrams (c) voltage of power supply, and (d) using the same power supply as calculated in (c), how long it will take in minutes if the device is not insulated so that heat loss is 400 kJ? 5 answers in BOXESarrow_forward
- Steam is contained in a 4-L volume at a pressure of 1.5 MPa and a temperature of 400 C. If the pressure is held constant by expanding the volume while 20 kJ of heat is added, the final temperature is nearestarrow_forwardA piston cylinder device initially contains 1.2 kg of a liquid-vapor mixture of water at 2 bars and 0.233 m3 the piston rests on stops and will not move until the system pressure reaches 10 bars. Heat is now transferred at a constant rate of 250 kJ/min. determine (a) the mass of initial liquid, (b) the heat added before the piston moves in kilojoules, and (c) the time required before the piston moves in minutesarrow_forwardA piston-cylinder device initially contains 0.9 m' of saturated water vapor at 350 kPa. At this state, the piston is resting on a set of stops, and the mass of the piston is such that a pressure of 400 kPa is required to move it. Heat is now slowly transferred to the steam until the volume doubles. Show the process on a P-V diagram with respect to saturation lines and determine (a) the final temperature, (b) the work done during this process, (c) the total heat transfersarrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





