
Concept explainers
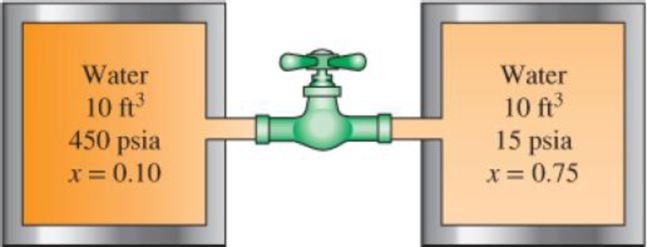
Two 10-ft3 adiabatic tanks are connected by a valve. Initially, one tank contains water at 450 psia with 10 percent quality, while the second contains water at 15 psia with 75 percent quality. The valve is now opened, allowing the water vapor from the high-pressure tank to move to the low-pressure tank until the pressure in the two becomes equal. Determine the final pressure and the final mass in each tank.
FIGURE P4–142E
The final pressure of each tank.
The final mass of each tank.
Answer to Problem 142RP
The final pressure of each tank is
The final mass of each tank is
Explanation of Solution
Write the expression for the energy balance equation.
Here, the total energy entering the system is
Simplify Equation (V) and write energy balance relation of two adiabatic tanks.
Here, the heat to be transfer into the system is
Substitute 0 for
Here, the initial mass of tank A is
Write the expression for initial mass of tank A.
Here, the volume of the tank A is
Write the expression for initial mass of tank B.
Here, the volume of the tank B is
Write the expression for total mass of tank.
Write the expression for initial total internal energy contained in both tanks.
Write the expression for initial is equal to final specific internal energy of tank.
Determine the total volume of both the tanks.
Write the expression for initial is equal to final specific volume of tank.
Write the expression for final mass contained in both tanks.
Conclusion:
At initial pressure and quality of initial state for tank A as 450 psia and 0.10, find the value of initial specific volume and specific internal energy of the tank.
Here, the specific volume of saturated liquid for tank A is
Here, the specific internal energy of saturated liquid for tank A is
Substitute
Substitute
At initial pressure and quality of initial state for tank B as 15 psia and 0.75, find the value of initial specific volume and specific internal energy of the tank.
Here, the specific volume of saturated liquid for tank B is
Here, the specific internal energy of saturated liquid for tank B is
Substitute
Substitute
Substitute
Substitute
Substitute
Substitute
Substitute
Substitute
Substitute
By above calculation from Table A-5E “saturated water” the final pressure of both tanks as
Thus, the final pressure of each tank is
Substitute
Thus, the final mass of each tank is
Want to see more full solutions like this?
Chapter 4 Solutions
Thermodynamics: An Engineering Approach
- A 0.3 m³ rigid tank contains refrigerant 134a at 650 kPa. Initially, 45% of the volume is occupied by liquid R-134a and the rest is occupied by vapor. When a valve at the bottom of the tank is opened, saturated liquid refrigerant leaves the tank. The valve is closed when tank is only occupied by saturated vapor. Heat is transferred to the tank to keep the temperature constant. How much refrigerant leaves the tank? What is the amount of heat transferred to the system? kg KJarrow_forwardAn insulated, vertical piston–cylinder device is purchased from a manufacturer. Upon purchase, it contains 3 kg of water at precisely 20 degrees celcius. The mass of the piston is such that it maintains a constant pressure of 300 kPa inside the cylinder. Liquid water at 2 MPA and 873.15 Kelvin is allowed to enter the cylinder from a supply line until the instant that the last drop of liquid in the cylinder vaporizes (saturated vapor). Find the mass of the steam that has entered [kg].arrow_forwardInitially, 200 L of saturated vapor refrigerant-134a in a piston. The piston is free to move, and its mass is such that it maintains a pressure of 900 kPa on the refrigerant. The refrigerant is now heated until its temperature rises to 60°C. The work done during this process is Select one: 73.55 kJ 37.55 kJ 27.49 kJ O 72.49 kJarrow_forward
- 2. A frictionless piston-cylinder device initially contains 50 L of saturated liquid refrigerant-RI34a. The piston is free to move, and its mass is such that it maintains a pressure of 500 kPa on the refrigerant. The refrigerant is now heated until its temperature rises to 70°C. Calculate the work done during this process. R134a P=constantarrow_forwardTwo kilograms of steam is compressed at constant pressure in a piston/cylinder from an initial state of 500 kPa and 300°C to a saturated vapor. Determine the work for the process.arrow_forwardWater is heated in a piston/cylinder arrangement at constant pressure. Initially the water is saturated vapor at 200 kPa and has a specific volume of 0.88578 m/kg. At the final state the temperature of the water is 300 C and has a specific volume of 1.31623 m/kg. The mass of the water is 2 kg. In the question that follows, select the answer that is closest to the true value. What is the work done by the water in units of kJ ?arrow_forward
- Water initially at 300 kPa and 0.5 m³/kg is contained in a piston-cylinder device fitted with stops so that the water supports the weight of the piston and the force of the atmosphere. The water is heated until it reaches the saturated vapor state and the piston rests against the stops. With the piston against the stops, the water is further heated until the pressure is 600 kPa. On the P-vand T-v diagrams, sketch, with respect to the saturation lines, the process curves passing through both the initial and final states of the water. Label the states on the process as 1, 2, and 3. On both the P-vand T-v diagrams, sketch the isotherms passing through the states and show their values, in °C, on the isotherms. Solve this using appropriate software. Use data from the tables. Water 300 kPa 0.5 m²/kg (Please upload your response/solution using the controls below.)arrow_forwardSubcooled water at 5°C is pressurised to 350 kPa with no increase in temperature, and then passed through a heat exchanger where it is heated until it reaches saturated liquid-vapour state at a quality of 0.63. If the water absorbs 499 kW of heat from the heat exchanger to reach this state, calculate how many kilogrammes of water flow through the pipe in an hour. Give your answer to one decimal place. Please provide correct solution Don't copypaste in cheggarrow_forwardA 0.78 m³ tank initially contains water at 260°C and a quality of 0.81. A valve at the top of the tank is opened and allows for saturated vapor at 260°C to leave the tank. The tank is maintained isothermally during this process until the tank is filled with only saturated vapor at 260⁰ C. What is the mass of steam that leaves the tank during this process? How much heated is added to the tank during this process. KJ kgarrow_forward
- A piston-cylinder device whose piston is resting on top of a set of stops initially contains 0.6 kg of helium gas at 100 kPa and 25°C. The mass of the piston is such that 500 kPa of pressure is required to raise it. How much heat must be transferred to helium before the piston starts rising? The specific heat of helium at room temperature is cy= 3.1156 kJ/kg-K (Table A-2). The amount of heat to be transferred to helium is kJ.arrow_forwardASAParrow_forwardIn a piston cylinder Assembly, R-134a refrigerant is available in 240 kPa and 40°C at a volume of 0.1 m3. The pressure created by the piston, which is held constant with the pin, and the masses and atmospheric pressure on it, is 280 KPA. It is heated in constant volume until the pressure inside the cylinder is increased to 280 KPA. And then the heat is drawn by a slow process with the cooling process by pulling the pin. If the temperature reached after this process is 50°C, find heat and work interactions during these two processes.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





