
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18, Problem 18.57SP
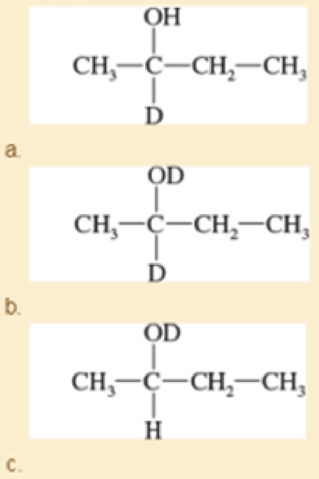
Both NaBH4 and NaBD4 are commercially available, and D2O is common and inexpensive. Show how you would synthesize the following labeled compounds, starting with butan-2-one.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please be clear in your writing, solve step by step
Compound A (C9H12), when hydrogenated by catalysis on Pd / C, absorbs 3 equivalents of H2 to give compound B (C9H18). Ozonolysis of compound A gives cyclohexanone (C6H10O). Compound A reacting with NaNH2 / NH3 followed by addition of CH3Br gave compound C (C10H14). What are the structures of compounds A, B, and C?
Provide the structure of the missing products or
reactants. Show the relevant stereochemistry if
necessary.
H3C
Br
H₂O
_SH
[4©
1 product
2 products (2 isomers)
✓ OH + ?<
Devise a route to convert propene to propanone.
Specify, in order, the reagents you would use. The reaction may require more than one step. If there is more than one feasible route, show only one.
a NBS
b H₂, Pd
c H₂O, H₂SO4
d PCC
e Br₂ in H₂O
f K₂Cr₂O7, H₂SO4,
H₂O, heat
g Cl₂, heat or light
h Cl2
i HCI
j NaOH
k OsO4/H₂O2
1. BH3
2. H₂O2, NaOH
m SOCI2₂, pyridine
I
n
Specify reagents using letters from the table, first reagent on the left. Example: ab
The reagents are (in order of use):
HBr
o Br2,
Chapter 18 Solutions
Organic Chemistry (9th Edition)
Ch. 18.3 - Give the IUPAC name and (if possible) a common...Ch. 18.5D - NMR spectra for two compounds are given here,...Ch. 18.5D - Why were no products from the McLafferty...Ch. 18.5D - Use equations to show the fragmentation leading to...Ch. 18.5E - Prob. 18.5PCh. 18.7D - Show how you would synthesize each compound from...Ch. 18.8 - Prob. 18.7PCh. 18.9 - Predict the products of the following reactions....Ch. 18.9 - Show how the following transformations may be...Ch. 18.10 - Prob. 18.10P
Ch. 18.11 - Show how you would accomplish the following...Ch. 18.11 - Prob. 18.12PCh. 18.12 - Propose mechanisms for a. the acid-catalyzed...Ch. 18.12 - Rank the following compounds in order of...Ch. 18.13 - Prob. 18.15PCh. 18.13 - Show how you would accomplish the following...Ch. 18.14 - Prob. 18.17PCh. 18.14 - Prob. 18.18PCh. 18.14 - Prob. 18.19PCh. 18.14 - Prob. 18.20PCh. 18.15 - 2,4-Dinitrophenylhydrazine is frequently used for...Ch. 18.15 - Prob. 18.22PCh. 18.15 - Prob. 18.23PCh. 18.16 - Prob. 18.24PCh. 18.16 - Prob. 18.25PCh. 18.16 - Show what alcohols and carbonyl compounds give the...Ch. 18.16 - In the mechanism for acetal hydrolysis shown, the...Ch. 18.16 - Prob. 18.28PCh. 18.17 - Show how you would accomplish the following...Ch. 18.18 - Prob. 18.30PCh. 18.18 - Prob. 18.31PCh. 18.18 - Prob. 18.32PCh. 18.18 - Show how Wittig reactions might be used to...Ch. 18.19 - Predict the major products of the following...Ch. 18.20C - Prob. 18.35PCh. 18.20C - Predict the major products of the following...Ch. 18 - Draw structures of the following derivatives. a....Ch. 18 - Prob. 18.38SPCh. 18 - Predict the major products of the following...Ch. 18 - Rank the following carbonyl compounds in order of...Ch. 18 - Acetals can serve as protecting groups for...Ch. 18 - Sketch the expected proton NMR spectrum of...Ch. 18 - A compound of formula C6H10O2 shows only two...Ch. 18 - The proton NMR spectrum of a compound of formula...Ch. 18 - The following compounds undergo McLafferty...Ch. 18 - An unknown compound gives a molecular ion of m/z...Ch. 18 - Show how you would accomplish the following...Ch. 18 - Prob. 18.48SPCh. 18 - Prob. 18.49SPCh. 18 - Propose mechanisms for the following reactions.Ch. 18 - Show how you would accomplish the following...Ch. 18 - Show how you would synthesize the following...Ch. 18 - Predict the products formed when cyclohexanone...Ch. 18 - Predict the products formed when...Ch. 18 - Show how you would synthesize octan-2-one from...Ch. 18 - Prob. 18.56SPCh. 18 - Both NaBH4 and NaBD4 are commercially available,...Ch. 18 - When LiAIH4 reduces 3-methylcyclopentanone, the...Ch. 18 - Prob. 18.59SPCh. 18 - Show how you would accomplish the following...Ch. 18 - There are three dioxane isomers 1,2-dioxane,...Ch. 18 - Two structures for the sugar glucose are shown on...Ch. 18 - Prob. 18.63SPCh. 18 - Prob. 18.64SPCh. 18 - Prob. 18.65SPCh. 18 - Prob. 18.66SPCh. 18 - Within each set of structures, indicate which will...Ch. 18 - Prob. 18.68SPCh. 18 - Prob. 18.69SPCh. 18 - Prob. 18.70SPCh. 18 - The UV spectrum of an unknown compound shows...Ch. 18 - a. Simple aminoacetals hydrolyze quickly and...Ch. 18 - The mass spectrum of unknown compound A shows a...Ch. 18 - Prob. 18.74SPCh. 18 - Prob. 18.75SPCh. 18 - Prob. 18.76SPCh. 18 - Prob. 18.77SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardH₂C ཏཱཏི 1 ནི OH 1. Br2, PBг3 2. H₂O H3C OH Br The a-bromination of carbonyl compounds by Br2 in acetic acid is limited to aldehydes and ketones because acids, esters, and amides don't enolize to a sufficient extent. Carboxylic acids, however, can be a-brominated by first converting the carboxylic acid to an acid bromide by treatment with PBr3. Following enolization of the acid bromide, Br2 reacts in an a-substitution reaction. Hydrolysis of the acid bromide completes the reaction. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions :0: H3C Br Br + :::OH2 Br H₂O H3C Br заarrow_forwardAmantadine is effective in preventing infections caused by the influenza A virus and in treating established illnesses. It is thought to block a late stage in the assembly of the virus. Amantadine is synthesized by treating 1-bromoadamantane with acetonitrile in sulfuric acid to give N-adamantylacetamide, which is then converted to amantadine. CH,C=N in H,SO, Br NHCCH3 NH2 1-Bromoadamantane Amantadine (a) Propose a mechanism for the transformation in Step 1. (b) Describe experimental conditions to bring about Step 2.arrow_forward
- Consider the following transformation. Which reagent would give you the indicated product? S. O NalO4 O 1: XS LAH (LIAIH4), 2: H20 O XS H202 O N2OH, H20, Br2 O DMSO, C202C12 (Swern)arrow_forwardIndicate the products X and Y that are obtained in the following reactions. Briefly comment on each reaction. CH2C6H5 CH3 + AcOH + H₂O → X X + CH3-CH=P(C6H5)3 -> Yarrow_forwardWhat is the IUPAC name of the following salt? K* CH3CO-arrow_forward
- When (CH3CH2)3CBr is added to CH3OH at room temperature, the product is (CH3O)C(CH2CH3)3. Propose a mechanism(s) for the reactions leading to these products and used curved arrows to show the movement of electrons.arrow_forwardTreatment of a hydrocarbon A (molecular formula C9H18) with Br2 in the presence of light forms alkyl halides B and C, both having molecular formula C9H17Br. Reaction of either B or C with KOC(CH3)3 forms compound D (C9H16) as the major product. Ozonolysis of D forms cyclohexanone and acetone. Identify the structures of A–D.arrow_forwardWhen (CH3CH2)3CBr is added to CH3OH at room temperature, the product is(CH3O)C(CH2CH3)3. Propose a mechanism(s) for the reactions leading to theseproducts and use curved arrows to show the movement of electrons.arrow_forward
- 3 Treatment of 1-aminoadamantane, C„H„N, with methyl 2,4-dibromobutanoate in the presence of a nonnucleophilic base, R,N, involves two successive S,2 reactions and gives compound A. Propose a structural formula for compound A. R&N NH, + Br. C15H93NO, + 2 R,NH Br OCH Br 1-Aminoadamantane Methyl 2,4-dibromobutanoate Aarrow_forwardAside from the specified main starting material, only the following compounds can be used as sources of the carbon backbone for the target compound: acetaldehyde ethanol propanoic acid acetic acid ethyl bromoacetate propan-1-ol benzene (2E,4E)-hexa-2,4-diene propan-2-ol buta-1,3-diene methanol propan-2-one (acetone) (chloromethyl)benzene (benzyl chloride) 5-methyloxolan-2-one sodium cyanide 1-(chloromethyl)-4-methylbenzene phenylacetaldehyde sodium methoxide diethyl propanedioate (diethyl malonate) piperidine sodium ethoxide There are no restrictions with regards to the use of other reagents such as catalysts, solvents, redox agents, protection/deprotection agents, etc. as long as they will not be used as sources of carbon for the skeleton of the target compound. Design a retrosynthetic map and from it, propose a multi-step synthetic approach for the preparation of the target compound from the specified starting material. USE…arrow_forwardAside from the specified main starting material, only the following compounds can be used as sources of the carbon backbone for the target compound: acetaldehyde ethanol propanoic acid acetic acid ethyl bromoacetate propan-1-ol benzene (2E,4E)-hexa-2,4-diene propan-2-ol buta-1,3-diene methanol propan-2-one (acetone) (chloromethyl)benzene (benzyl chloride) 5-methyloxolan-2-one sodium cyanide 1-(chloromethyl)-4-methylbenzene phenylacetaldehyde sodium methoxide diethyl propanedioate (diethyl malonate) piperidine sodium ethoxide - There are no restrictions with regards to the use of other reagents such as catalysts, solvents, redox agents, protection/deprotection agents, etc. as long as they will not be used as sources of carbon for the skeleton of the target compound. 1. Design a retrosynthetic map and from it, propose a multi-step synthetic approach for the preparation of the target compound from the specified starting material.…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY