
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18.18, Problem 18.33P
Show how Wittig reactions might be used to synthesize the following compounds. In each case, start with an
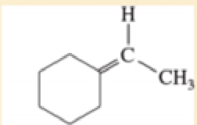
- a. Ph-CH=C(CH3)2
- b. Ph-C(CH3)=CH2
- c. Ph CH=CH-CH=CH-Ph
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1.
Write the structure of the major organic product for reactions a, b and e?
H₂C
b. H₂c-
CH,
CH₂
Cold KMnO,
OH
A KMnO,
H
c. CH3CH₂-CH=CH-CH₂CH3 + H₂SO4 →
Give the product.
NO ₂
0
HNO ₂
H₂SO4
Bromo cyclopentadiene reacts to form a Witting Reagent (A), but does not react with aldehydes
or ketones to give an alkene. Draw A and explain why it does not react with a carbonyl.
Br
1. PPh3
2. Base
A
R H
No Rx
Chapter 18 Solutions
Organic Chemistry (9th Edition)
Ch. 18.3 - Give the IUPAC name and (if possible) a common...Ch. 18.5D - NMR spectra for two compounds are given here,...Ch. 18.5D - Why were no products from the McLafferty...Ch. 18.5D - Use equations to show the fragmentation leading to...Ch. 18.5E - Prob. 18.5PCh. 18.7D - Show how you would synthesize each compound from...Ch. 18.8 - Prob. 18.7PCh. 18.9 - Predict the products of the following reactions....Ch. 18.9 - Show how the following transformations may be...Ch. 18.10 - Prob. 18.10P
Ch. 18.11 - Show how you would accomplish the following...Ch. 18.11 - Prob. 18.12PCh. 18.12 - Propose mechanisms for a. the acid-catalyzed...Ch. 18.12 - Rank the following compounds in order of...Ch. 18.13 - Prob. 18.15PCh. 18.13 - Show how you would accomplish the following...Ch. 18.14 - Prob. 18.17PCh. 18.14 - Prob. 18.18PCh. 18.14 - Prob. 18.19PCh. 18.14 - Prob. 18.20PCh. 18.15 - 2,4-Dinitrophenylhydrazine is frequently used for...Ch. 18.15 - Prob. 18.22PCh. 18.15 - Prob. 18.23PCh. 18.16 - Prob. 18.24PCh. 18.16 - Prob. 18.25PCh. 18.16 - Show what alcohols and carbonyl compounds give the...Ch. 18.16 - In the mechanism for acetal hydrolysis shown, the...Ch. 18.16 - Prob. 18.28PCh. 18.17 - Show how you would accomplish the following...Ch. 18.18 - Prob. 18.30PCh. 18.18 - Prob. 18.31PCh. 18.18 - Prob. 18.32PCh. 18.18 - Show how Wittig reactions might be used to...Ch. 18.19 - Predict the major products of the following...Ch. 18.20C - Prob. 18.35PCh. 18.20C - Predict the major products of the following...Ch. 18 - Draw structures of the following derivatives. a....Ch. 18 - Prob. 18.38SPCh. 18 - Predict the major products of the following...Ch. 18 - Rank the following carbonyl compounds in order of...Ch. 18 - Acetals can serve as protecting groups for...Ch. 18 - Sketch the expected proton NMR spectrum of...Ch. 18 - A compound of formula C6H10O2 shows only two...Ch. 18 - The proton NMR spectrum of a compound of formula...Ch. 18 - The following compounds undergo McLafferty...Ch. 18 - An unknown compound gives a molecular ion of m/z...Ch. 18 - Show how you would accomplish the following...Ch. 18 - Prob. 18.48SPCh. 18 - Prob. 18.49SPCh. 18 - Propose mechanisms for the following reactions.Ch. 18 - Show how you would accomplish the following...Ch. 18 - Show how you would synthesize the following...Ch. 18 - Predict the products formed when cyclohexanone...Ch. 18 - Predict the products formed when...Ch. 18 - Show how you would synthesize octan-2-one from...Ch. 18 - Prob. 18.56SPCh. 18 - Both NaBH4 and NaBD4 are commercially available,...Ch. 18 - When LiAIH4 reduces 3-methylcyclopentanone, the...Ch. 18 - Prob. 18.59SPCh. 18 - Show how you would accomplish the following...Ch. 18 - There are three dioxane isomers 1,2-dioxane,...Ch. 18 - Two structures for the sugar glucose are shown on...Ch. 18 - Prob. 18.63SPCh. 18 - Prob. 18.64SPCh. 18 - Prob. 18.65SPCh. 18 - Prob. 18.66SPCh. 18 - Within each set of structures, indicate which will...Ch. 18 - Prob. 18.68SPCh. 18 - Prob. 18.69SPCh. 18 - Prob. 18.70SPCh. 18 - The UV spectrum of an unknown compound shows...Ch. 18 - a. Simple aminoacetals hydrolyze quickly and...Ch. 18 - The mass spectrum of unknown compound A shows a...Ch. 18 - Prob. 18.74SPCh. 18 - Prob. 18.75SPCh. 18 - Prob. 18.76SPCh. 18 - Prob. 18.77SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is(are) the product(s) of the following reaction? H2SO, CH EIOH E. D. OE OEt OH CH B. C. A+B A. он CH OEt OH CHa OE! CH OA OEarrow_forwardA. REACTIONS OF ALKENES- provide the product(s) or reagents that are needed to accomplish the following transformations CH3 1. BH 3, THF 2. H₂O₂, NaOH, H₂Oarrow_forwardWhat is the major organic product obtained from the following reaction? A. CI B. H 1. (CH3CH₂)2CuLi 2. H3O+ C. D. •ay.a OHarrow_forward
- Which is the product of the following reaction sequence? 1. Br₂ 2. Excess NaNH, A 3. H₂O 4. HgSO4/H₂SO4/H₂O B с OH J D Harrow_forward8. What is the major organic product obtained from the following reaction? CH3 H₂C CH3 1. Hg(OAc)₂ H₂O 2. NaBH4arrow_forwardChoose the most appropriate reagent(s) for the synthesis of 1-butyne from ethyne. reagent(s) H = H но A 1. NANH2 B 1. NANH, 2. CH,CH3 2. CH,CH,OH CH,CH,OH D CH,CH,Br NaNH2arrow_forward
- OH 1. Hg(OAc)2, H20 2. NaBH4 .CH3 CH3 H2C H3C Acid-catalyzed addition of water to an alkene yields an alcohol with Markovnikov regiochemistry. The electrophilic H* adds to the sp2 carbon with the most hydrogens to yield the most stable carbocation intermediate, which then adds water to give the product alcohol. Because a carbocation intermediate is formed, rearrangements can occur prior to the addition of water. To avoid the possibility of rearrangement and still give a Markovnikov alcohol, alkenes can instead be treated with mercury(II) acetate in aqueous THF and then subsequently reduced with sodium borohydride. This reaction proceeds through a cyclic mercurinium ion intermediate which cannot rearrange. Water adds to the cyclic intermediate at the most substituted carbon to give an organomercury alcohol. The reduction step with sodium borohydride is complex and involves radicals. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing…arrow_forwardH. An ester is produced by the reaction of an alcohol with a carboxylic acid. For each ester below, underline the alcohol part and circle the carboxylic acid part. Then, name each ester. CHỊCH,CH,CH,CH,-O-C-CH,CH,CH, H CH₂ CH₂ CH 1 CH3arrow_forward5. Write the products for the following two reactions of pheno 1. NaOH 2. СН,CH Br HO 1. (CF,CO2) Hg /H2C-CH2 2. NaBH4arrow_forward
- 15. What is the main product of this reaction? A. B. C. OAC "CH 3 OAC OAC CH3 QAC CH3 CH3 NaOAc DMF OACarrow_forwardWhat is the best answer for the following reaction? OH Br H. A. 1) HO OH H,o 3) H,o 2) Mg, Diethylether B. 1) Mg, Diethylether 2) H30 3) HO OH H,o* с. 1) но OH H3O 2) Mg, Diethylether D. 1) Mg, Diethylether 2) HO онarrow_forwardWhat is/are the major product(s) of this reaction se 1. ВНЗ 2. H2O2, NaOH 3. PBR3 4. (CH3)3COK/(CH3)3COH +1arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY