
Concept explainers
a)
Interpretation:
Number of carbon-carbon sigma bonds present in the given benzene has to be identified.
Concept introduction:
Sigma bonds: A sigma bond is a covalent bond formed by head on overlap of atomic orbitals that carries two electrons. The bonding represented as a single line between two atoms.
Head-on overlap:
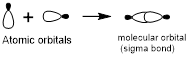
Head-on overlap of two atomic orbitals forms a molecular orbital known as sigma bonds.
Side-to-side overlap:
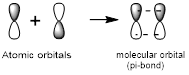
Side-to-side overlap of two atomic orbitals forms a molecular orbital known as pi-bonds.
b)
Interpretation:
Number of carbon-carbon sigma bonds present in the given cyclobutane has to be identified.
Concept introduction:
Sigma bonds: A sigma bond is a covalent bond formed by head on overlap of atomic orbitals that carries two electrons. The bonding represented as a single line between two atoms.
Head-on overlap:
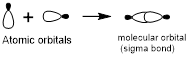
Head-on overlap of two atomic orbitals forms a molecular orbital known as sigma bonds.
Side-to-side overlap:
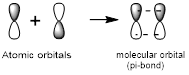
Side-to-side overlap of two atomic orbitals forms a molecular orbital known as pi-bonds.
c)
Interpretation:
Number of carbon-carbon sigma bonds present in the given 3-ethyl-2-methylpentane has to be identified.
Concept introduction:
Sigma bonds: A sigma bond is a covalent bond formed by head on overlap of atomic orbitals that carries two electrons. The bonding represented as a single line between two atoms.
Head-on overlap:
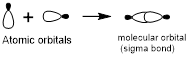
Head-on overlap of two atomic orbitals forms a molecular orbital known as sigma bonds.
Side-to-side overlap:
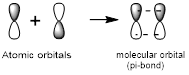
Side-to-side overlap of two atomic orbitals forms a molecular orbital known as pi-bonds.
Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
Chemistry
- Give the IUPAC names of structures containing two carbon atoms for the following classes of compounds: (a) ether: (b) aldehyde: (c) carboxylic acid: (d) ester:arrow_forwardWhat is the molecular formula for a 5-carbon hydrocarbon with one pie bond and one ring?arrow_forwardwrite the structure formulas of alkanes with molecular formula C6H14, which with chlorine give: a) three monochlorinated isomers? b) five monochlorinated isomers c) only two monochlorinated isomersarrow_forward
- Classify the following hydrocarbons, and draw a Lewis structure for each one. A compound may fit into more than one of the following classifications: alkane cycloalkane aromatic hydrocarbon alkene cycloalkene alkyne cycloalkyne CH,-C=C-CH CH, —CH, —CH, —сH (d) CHCH, (f) CCH - СНC(CH), (h) - CH2CH3 (i)arrow_forwardTRUE OR FALSE (a) Both ethylene and acetylene are planar molecules. (b) An alkene in which each carbon of the double bond has two different groups bonded to it will show cis-trans isomerism. (c) Cis-trans isomers have the same molecular formula but a different connectivity of their atoms. (d) Cis-2-butene and trans -2-butene can be interconverted by rotation about the carbon–carbon double bond. (e) Cis-trans isomerism is possible only among appropriately substituted alkenes. (f) Both 2-hexene and 3-hexene can exist as pairs of cis-trans isomers. (g) Cyclohexene can exist as a pair of cis-trans isomers. (h) 1-Chloropropene can exist as a pair of cis-trans isomers.arrow_forwardThe following is a structural diagram for penicillin G, an antibiotic compound with outstanding antibacterial activity. It is obtained from the liquid filtrate of molds. HO H H H C-C N- H Penicillin G can be described as an Organic Co -N- organic inorganic CH₂ H CH₂ -COOH compound. One functional group found in penicillin G is the ◆ grouparrow_forward
- Give the molecular formula of a hydrocarbon containing six carbon atoms that is an aromatic hydrocarbon.arrow_forwardA certain hydrocarbon has a molecular formula of C5H8. Which of the following is not a structural possibility for this hydrocarbon? (a) It is a cycloalkane. (b) It contains one ring and one double bond. (c) It contains two double bonds and no rings. (d) It is an alkyne.arrow_forwardHow many asymmetric carbons are present in Ethyl-2,2,4-tnmethypentane? Indicate them.arrow_forward
- Give the IUPAC names of structures containing two carbon atoms for the following classes of compounds: (a) alkene: (b) alkyne: (c) alkyl halide: d) alcohol:arrow_forward(a) Alkenes are relatively stable compounds but are more reactive than alkanes and serve as a feedstock for the petrochemical industry because they can participate in a wide variety of reactions. Predict the products obtained from following reaction. Write the chemical reaction and name the products according to IUPAC system. (i) the reaction of 3-ethyl-3-methyl-1-pentene with hydrogen bromide (ii) the reaction of 3-ethyl-2-pentene with hydrogen bromidearrow_forward1-Propylamine, propan-1-ol, acetic acid, and butane have about the same molar masses. Which would you expect to have the (a) highest boiling point, (b) lowest boiling point, (c) least solubility in water, and (d) least chemical reactivity? Have each member of your group chose a part to answer, andthen discuss with each other why those answers were chosen.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

