
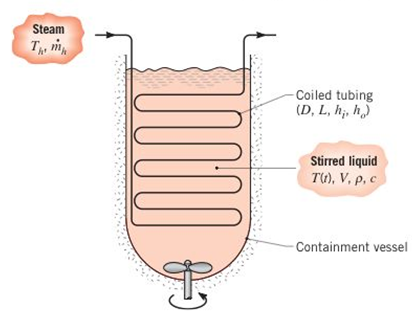
Batch processes are often used in chemical and pharmaceutical operations to achieve a desired chemical composition for the final product and typically involve a transient heating operation to take the product from room temperature to the desired process temperature. Consider a situation for which a chemical of density
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Introduction to Heat Transfer
- Assume the following: ●In a light water reactor, approximately 1.0kg of spent nuclear fuel is produced for every50. megawatt-days (MWd) of thermal energy input. ●For the purposes of this analysis, assume the spent nuclear fuel produced is 1.8% Pu-240by mass. Pu-240 has a half-life of 6,430 years. ●The thermal efficiency of the power plant is 38% ●Use equation for radioactive decay: Qt= Q0^e-kt where: ○Qt = quantity of radioactive material at time t ○Q0 = original quantity of radioactive material ○k = the decay constant ○t = time interval in years ○The decay constant k is defined by the relation: Half life = ln(2)/k Diablo Canyon stores 95% of its spent nuclear fuel onsite. ii) How many metric tons of Pu-240 in the spent fuel will still be onsite in that year?arrow_forwardTwo ideal gases were delivered to two containers of equal volumes temperature and pressure. One of the containers has a fixed volume, while the other is a cylinder equipped with a weightless piece of furniture. initially the pressure of gas equals blood pressure. Then the gases are heated with a Bunsen burner. Draw a picture to represent the processes described and determine what are the signs of q and w of the gases under these conditions? will exist difference in the internal energy of these systems? your job.arrow_forwardQ1: Ammonia is absorbed in water from a mixture with air using a column operating at atmospheric pressure and 295 K. The resistance to transfer can be regarded as lying entirely within the gas phase. At a point in the column, the partial pressure of the ammonia is 6.6 kN/m The back pressure at the water interface is negligible, and the resistance to transfer may be regarded as lying in a stationary gas film 1 mm thick. If the diffusivity of ammonia in air is 0.236 cm2 /s, what is the transfer rate per unit area at that point in the column?arrow_forward
- 3. Mass Transfer from a Pipe Wall. Air at 52.6 °C and 1 atm abs pressure passes through a pipe with naphthalene lining. The pipe's diameter in 25 mm. The vapor pressure of naphthalene at 52.6 °C is 1.0 mm Hg. The diffusivity in air of naphthalene at 0 °C is 5.16x106 m?/s. For a 0.20 m section of a pipe that is lined with naphthalene, calculate the exiting concentration if the entering air carries 0.001 kgmol/m of naphthalene and has a velocity of 0.30 m/s. A.3-3 Physical Properties of Air at 101.325 kPa (1 Atm Abs), SI Units Ax 105 (Pa s, or (K) (kg/m) (kJ/kg K) kg/m s) (W{m K) N, (1/K) Bx 10 9Bp 1 (1/K -m) k (°C) - 17.8 255.4 1.379 2.79 x 10% 2.04 x 108 1.72 x 10 1.12 x 10 0.02925 0.702 2.95 0.775 x 10 0.03115 0.694 2.74 0.534 x 108 0.03323 0.692 2.54 0.386 x 10* 0.03531 0.689 2.38 0.289 x 108 0.03721 0.687 2.21 0.214 x 108 0.03894 0.686 2.09 0.168 x 10* 0.04084 0.684 1.98 0.130 x 10 0.04258 0.680 1.87 0.104 x 108 1.62 0.02250 0.720 3.92 0.02423 0.715 3.65 1.0048 273.2 1.293 1.0048…arrow_forward3.23 3.24 d 3.25. Gas at constant T and P is contained in a supply line connected through a valve to closed tank containing the same gas at a lower pressure. The valve is opened to allow flow of gas into the tank, and then is shut again. (a) Develop a general equation relating n1 and n2, the moles (or mass) of gas in the tank at the beginning and end of the process, to the properties U1 and U2, the internal energy of the gas in the tank at the beginning and end of the process, and H', the enthalpy of the gas in the supply line, and to Q, the heat transferred to the material in the tank during the process. (b) Reduce the general equation to its simplest form for the special case of an ideal gas with constant heat capacities. (c) Further reduce the equation of (b) for the case of n1 = 0. (d) Further reduce the equation of (c) for the case in which, in addition, Q = 0. (e) Treating nitrogen as an ideal gas for which Cp equation to the case in which a steady supply of nitrogen at 25°C and…arrow_forwardA container is filled with a mixture of 2 fluids having densities of 3 kg/L (fluid A) and 4000 kg/m^3 (fluid B). What will be the overall density (in g/cm^3) of the mixture of 1/4 of the container is filled with fluid A and 1/2 is filled with fluid B?arrow_forward
- 4. One mole of helium gas is injected into each side of a slidable, airtight lead piston that separates two chambers of a sealed cylinder. The outside of the cylinder is insulated everywhere except where noted below. The cylinder and piston have lengths and cross- sectional area labeled in the diagram. The helium in the left chamber is heated from outside at a rate of 450 W, and the helium in the other chamber expels heat into a cool region. The full system eventually comes to steady-state. When it reaches steady state, the force needed to hold on the end cap is measured to be 18,000 N, and the piston has slid to an equilibrium point that is a distance à from the left end. force holding cap = 18,000N pressure. cross-sectional area of cylinder = 0.02m² x = ? Imol He do dt = +450W 45cm 15cm lead piston a. Find the temperature difference of the two chambers. b. Find the distance x. dQ dt Assume helium behaves as an ideal gas, and that heat transfer through the container walls is…arrow_forwardHow does the presence of chemical reactions affect phase equilibrium in thermodynamic systems within the field of mechanical engineering?arrow_forwardThere is a heated continuous stirred tank reactor shown below where the density is constant through out the system. On the other hand the volume of reaction mixture may vary. The thermal capacitance of the coil used to heat the tank is assumed to be negligible. There is a 2nd order elementary chemical reaction 2A → C taking place in the reactor. Cai Tai qa 2A ->C v „ro Tc Ac In the figure shown above Cai, Tai,qa is the inlet concentration of component A , inlet stream temperature and inlet volumetric flow rate, V is the volume of the reaction mixture, ro is the density through out the system. Tc, Ac and U are the coil temperature, surface area of the coil and heat transfer coefficient of the coil. Cc, T and Ca are the concentration of product C, temperature of the reactor and exit concentration of component A, qc is the outlet volumetric flowrate. The rate constant is assumed to be independent of temperature denoted as ko. The heat of reaction is denoted as AHrxn. Cp is the heat…arrow_forward
- NOTE: Complete solution and show the derivation/cancellation of units. N m An ideal gas has a gas constant of 319.8 kg K and a gas ratio of 1.26. Calculate the heat rejected per kg of gas: A. when the gas is contained in a rigid vessel at 3 bar and 315°C and then cooled until the pressure falls to 1.5 bar. B. when the gas enters a pipeline at 280°C and flows steadily at constant pressure to the end of the pipe where the temperature is 20°C.arrow_forwardWhen a sheet metal is suddenly removed from an oven and exposed to the environment, it cools due to loss due to convection. The rate at which the plate temperature T varies with time is given by: A, -[ h(T– T„)] pVC, dT %3D dt where As is the surface area of the plate, p = 300 kg / m³ is its mass density, V is its volume, CV= 900 J/ kg / K is your specific heat in the volume condition constant. In addition, h = 30 W / m² / K is the coefficient heat transfer and To is the temperature environment. Given the information above, obtain: a) The algebraic equation that represents the ODE described by the model using a power series. b) The T xt graph using 10 terms of the series, and compare the result with the solution analytical method obtained by the Laplace transform. Data: V = 0,003 m³; As = 0,25 m²; T(0) = 673 K; To = 298 Karrow_forward6. A mass of 0.20 kg of metal having a temperature of 100 deg. C is plunged into 0.04 kg of water at 20 deg. C The temperature of the water and metal becomes 48 deg. C. The latent heat of ice at 0 eg. C is 335 kJ/kg-K and the specific heat capacity of water is 4.19 kJ/kg-K. Assuming no heat loss to the surroundings, determine the specific heat capacity of the metal in kJ/kg-Karrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





