
Concept explainers
(a)
Interpretation:
The chemical name of
(b)
Interpretation:
The chemical name of
(c)
Interpretation:
The chemical name of
(d)
Interpretation:
The chemical name of
Concept introduction:
Ions contains bunch of atoms having covalent bonds and charged are called polyatomic ions. The name of certain polyatomic ions are given below
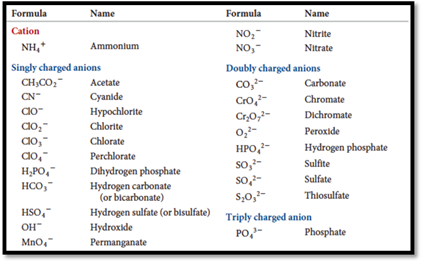
Figure 1
(e)
Interpretation:
The chemical name of
Concept introduction:
Ions contains bunch of atoms having covalent bonds and charged are called polyatomic ions. The name of certain polyatomic ions are given below
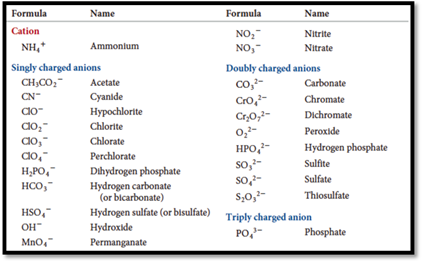
Figure 1
(f)
Interpretation:
The chemical name of
(g)
Interpretation:
The chemical name of
Concept introduction:
Ions contains bunch of atoms having covalent bonds and charged are called polyatomic ions. The name of certain polyatomic ions are given below

Figure 1
(h)
Interpretation:
The chemical name of
Concept introduction:
Ions contains bunch of atoms having covalent bonds and charged are called polyatomic ions. The name of certain polyatomic ions are given below

Figure 1
(i)
Interpretation:
The chemical name of
(j)
Interpretation:
The chemical name of
Concept introduction:
Ions contains bunch of atoms having covalent bonds and charged are called polyatomic ions. The name of certain polyatomic ions are given below

Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
General Chemistry: Atoms First
- The present average concentration (mass percent) of magnesium ions in seawater is 0.13%. A chemistry textbook estimates that if 1.00 × 108 tons Mg were taken out of the sea each year, it would take one million years for the Mg concentration to drop to 0.12%. Do sufficient calculations to either verify or refute this statement. Assume that Earth is a sphere with a diameter of 8000 mi, 67% of which is covered by oceans to a depth of 1 mi, and that no Mg is washed back into the oceans at any time.arrow_forwardGiven that the density of argon is 1.78 g/L under standard conditions of temperature and pressure, how many argon atoms are present in a room with dimensions 4.0 m 5.0 m 2.4 m that is filled with pure argon under these conditions of temperature and pressure?arrow_forwardChlorine exists mainly as two isotopes, 37Cl and 33Cl. Which is more abundant? How do you know?arrow_forward
- What is the mass of fish, in kilograms, that one would have to consume to obtain a fatal dose of mercury, if the fish contains 30 parts per million of mercury by weight? (Assume that all the mercury from the fish ends up as mercury (II) chloride in the body and that a fatal dose is 0.20 g of HgCl2.) How many pounds of fish is this?arrow_forward4-102 Aspartame, an artificial sweetener used as a sugar substitute in some foods and beverages, has the molecular formula C14H18N2O5. (a) How many mg of aspartame are present in 3.72 × 1026 molecules of aspartame? (b) Imagine you obtain 25.0 mL of aspartame, which is known to have a density of 1.35 g/mL. How many molecules of aspartame are present in this volume? (c) How many hydrogen atoms are present in 1.00 mg of aspartame? (d) Complete the skeletal structure of aspartame, where all the bonded atoms are shown but double bonds, triple bonds, and/or lone pairs are missing. (e) Identify the various types of geometries present in each central atom of aspartame using VSEPR theory. (f) Determine the various relative bond angles associated with each central atom of aspartame using VSEPR theory. (g) What is the most polar bond in aspartame? (h) Would you predict aspartame to be polar or nonpolar? (i) Is aspartame expected to possess resonance? Explain why or why not. (j) Consider the combustion of aspartame, which results in formation of NO2(g) as well as other expected products. Write a balanced chemical equation for this reaction. (k) Calculate the weight of C02(g) that can be prepared from 1.62 g of aspartame mixed with 2.11 g of oxygen gas.arrow_forward2. Classify each of the following as a binary ionic compound, ternary ionic compound, binary molecular compound, binary acid, or ternary oxyacid. (a) ammonia, NH3 (c) bleach, NaCIO охyan (a) CC (c) SC (b) iodized salt, KI 13. Provi (d) battery acid, H,SO4(aq) ions.arrow_forward
- Q4(B) Define mass number. Explain the following redox reactions.arrow_forwardA 1.30 g sample of titanium chemically combines with chlorine gas to form 5.16 g of titanium chloride. (a) What is the empirical formula of titanium chloride? (b) What is the percent by mass of titanium and the percent by mass of chloride in the sample?arrow_forwardA compound is composed of carbon, hydrogen, nitrogen and oxygen. When a 1.500-g sample of the compound is completely combusted, it yields 1.476 g of CO2and 0.605 g of H2O. In a separate analysis to determine nitrogen, 1.500 g of the compound is found to produce 0.313 g of N2. (a) Calculate the mass percent of each element in the compound. (b) Determine the empirical formula of the compound. (c) If the compound has a molar mass of 134 g/mol, what is the molecular formula?arrow_forward
- Most abundant metal on the surface of the earth:(a) Iron(b) Aluminium(c) Calcium(d) Sodiumarrow_forwardWrite the empirical formulas for (a) glucose, a substance also known as either blood sugar or dextrose—molecular formulaC6H12O6; (b) nitrous oxide, a substance used as an anesthetic and commonly called laughing gas—molecular formula N2O.arrow_forwardA gaseous binary compound has a vapor density that is 2.53 times that of nitrogen at 100°C and atmospheric pressure. When 8.21 g of the gas reacts with AlCl3 at 100°C, 1.62 g gaseous nitrogen is produced, removing all of the nitrogen originally present.(a) Estimate the molecular mass of the gaseous compound.(b) How many nitrogen atoms are there in a molecule ofthe compound?(c) What is the maximum possible value of the atomicmass of the second element?(d) Are other values possible for the atomic mass of thesecond element? Use a table of atomic masses to identify the element that best fits the data.(e) What is the molecular formula of the compound?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax





