
Concept explainers
(a)
Interpretation:
The type of intermolecular force present in Chloroform has to be given.
Concept introduction:
Intermolecular forces:
Intermolecular forces are like cohesive forces, acting between the molecules. The overall stability of a compound depends on how strong the molecules are held together. Intermolecular force is concerned about the overall stability of a substance. A stable substance has stronger intermolecular forces.
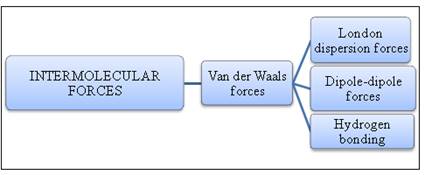
Figure 1
(b)
Interpretation:
The type of intermolecular force present in Oxygen has to be given.
Concept introduction:
Intermolecular forces:
Intermolecular forces are like cohesive forces, acting between the molecules. The overall stability of a compound depends on how strong the molecules are held together. Intermolecular force is concerned about the overall stability of a substance. A stable substance has stronger intermolecular forces.
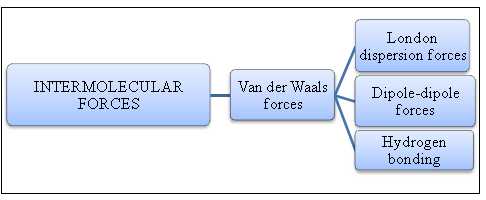
Figure 1
(c)
Interpretation:
The type of intermolecular force present in Polyethylene has to be given.
Concept introduction:
Intermolecular forces:
Intermolecular forces are like cohesive forces, acting between the molecules. The overall stability of a compound depends on how strong the molecules are held together. Intermolecular force is concerned about the overall stability of a substance. A stable substance has stronger intermolecular forces.

Figure 1
(d)
Interpretation:
The type of intermolecular force present in Methanol has to be given.
Concept introduction:
Intermolecular forces:
Intermolecular forces are like cohesive forces, acting between the molecules. The overall stability of a compound depends on how strong the molecules are held together. Intermolecular force is concerned about the overall stability of a substance. A stable substance has stronger intermolecular forces.

Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
General Chemistry: Atoms First
- Explain why liquids assume the shape of any container into which they are poured, whereas solids are rigid and retain their shape.arrow_forwardWhat is the relationship between the structures of buckminsterfullerene and carbon nanotubes?arrow_forwardName the type(s) of intermolecular forces that exists between molecules (or basic units) in each of the following species: (a) NaCL (b) CS2arrow_forward
- Name the type(s) of intermolecular forces that exists between molecules (or basic units) in each of the following species and identify the following species that are capable of hydrogen-bonding among themselves. (a) BeH2, (b) CH3COOHarrow_forwardBased on the type or types of intermolecular forces, predictthe substance in each pair that has the higher boiling point:(a) propane (C3H8) or n-butane (C4H10), (b) diethyl ether(CH3CH2OCH2CH3) or 1-butanol (CH3CH2CH2CH2OH),(c) sulfur dioxide (SO2) or sulfur trioxide (SO3), (d) phosgene(Cl2CO) or formaldehyde (H2CO).arrow_forwardWhich type of intermolecular force accounts for each ofthese differences? (a) CH3OH boils at 65 °C; CH3SH boils at6 °C. (b) Xe is a liquid at atmospheric pressure and 120 K,whereas Ar is a gas under the same conditions. (c) Kr,atomic weight 84 amu, boils at 120.9 K, whereas Cl2, molecularweight about 71 amu, boils at 238 K. (d) Acetone boilsat 56 °C, whereas 2-methylpropane boils at -12 °C.arrow_forward
- Which of the following set of compounds form an ionic solid, a molecular solid, and a covalent network solid, in that order? (A) Na2O, Na2O2, SiO2; (B) Na2O, MgO, Al2O3; (C) BaO, BaO2, CO2; (D) CaO, SO2, SiO2;arrow_forwardName the type(s) of intermolecular forces that exists between molecules (or basic units) in each of the following species and identify the following species that are capable of hydrogen-bonding among themselves. (a) C2H6, (b) HI, (c) KF, (d) BeH2, (e) CH3COOHarrow_forwardThe substance with the lowest known boiling point (4 K) is helium, an atomic element that has two electrons. Hydrogen isa diatomic molecule and also has two electrons, but its boiling point is significantly higher, at 20.28 K.(a) What is the dominant intermolecular force between a pair of helium atoms and a pair of H2 molecules?(b) Why do you think H2 has a higher boiling point?arrow_forward
- Which of the following is FALSE about the water molecule? A Intermolecular forces among water molecules are the least extensive when it is in liquid form. The atoms comprising the water molecule are covalently bonded with one another. (c) The H atoms bear the partial positive charge. Its molecular geometry is not linear.arrow_forwardWhich of the following compounds exhibit London dispersion forces: (a) NH 3; (b) H 2O; (c) HCl; (d) ethane (C 2H 6)?arrow_forwardWhat types of intermolecular forces are present in each compound: (a) HCl; (b) C 2H 6 (ethane); (c) NH 3?arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning



