
Interpretation:
The radius of Tungsten in picometers has to be calculated.
Concept Introduction:
Body-centered unit cell:
A body – centered cubic cell is a type of unit cell with atoms arranged in all the vertices of the unit cell and one atom at the center of the cube.
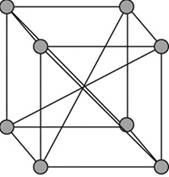
Figure 1
A body – centered cubic unit cell has its all the eight vertices arranged with atoms in it. Eight atoms are arranged in a simple cubic unit cell in eight vertices. Each atom in a vertex is shared by eight neighboring unit cells such that each cubic unit cell has
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
General Chemistry: Atoms First
- Explain why liquids assume the shape of any container into which they are poured, whereas solids are rigid and retain their shape.arrow_forwardLiquid ethylene glycol, HOCH2CH2OH, is one of the main ingredients in commercial antifreeze. Would you predict its viscosity to be greater or less than that of ethanol, CH3CH2OH?arrow_forwardIn terms of the kinetic molecular theory, in what ways are liquids similar to gases? In what ways are liquids different from gases?arrow_forward
- Why are capillary rises and depressions not seen for cylinders with large radii?arrow_forwardThe compounds ethanol (C2H5OH) and dimethyl ether (CH3OCH3) have the same molecular formula. Which is expected to have the higher surface tension? Why?arrow_forwardWhich of the following do you expect to be molecular solids? a silicon tetrachloride, SiCl4 b lithium bromide, LiBr c sodium fluoride, NaF d bromine chloride, BrClarrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax  Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





