
Concept explainers
(a)
Interpretation: The structural formula for 1,3-dichlorobenzene has to be drawn.
Concept introduction:
A benzene ring has six carbon atoms which are
The structure of the benzene derivative when one or more hydrogen atoms are replaced by other groups, can be drawn as follows,
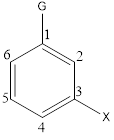
The systematic name for the benzene derivatives is written by taking the parent name
(b)
Interpretation: The structural formula for 1-bromo-4-methylbenzene has to be drawn.
Concept introduction: A benzene ring has six carbon atoms which are
The structure of the benzene derivative when one or more hydrogen atoms are replaced by other groups, can be drawn as follows,

The systematic name for the benzene derivatives is written by taking the parent name
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Chemistry & Chemical Reactivity
- Name or write the condensed structural formula for the following compounds:(a) 4-methyl-2-pentene (b) cis-2,5-dimethyl-3-hexenearrow_forwardDraw structures for the following molecules. (a) 2-iodo-2,3-dimethylheptane (b) 4-cyclopropyl-3-ethyl-2-methyloctane (c) 1-ethyl-2,4-dimethylcyclopentanearrow_forwardA certain hydrocarbon has a molecular formula of C5H8. Which of the following is not a structural possibility for this hydrocarbon? (a) It is a cycloalkane. (b) It contains one ring and one double bond. (c) It contains two double bonds and no rings. (d) It is an alkyne.arrow_forward
- (a) What structural feature is associated with each type of hydrocarbon: alkane, cycloalkane, alkene, and alkyne?(b) Give the general formula for each type.(c) Which hydrocarbons are considered saturated?arrow_forward5. Give the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group: Name the functional group:arrow_forwardA certain hydrocarbon has a molecular formula of C5H8. Which of the following is not a structural possibility for this hydrocarbon: (d) It contains an alkyne O It contains one ring and one double bond (c) It contains two double bonds and no rings O (b) It contains one ring and no double bondsarrow_forward
- Indicate whether each of the following molecules is capableof geometrical isomerism. For those that are, draw the structures:(a) 1,1-dichloro-1-butene, (b) 2,4-dichloro-2-butene,(c) 1,4-dichlorobenzene, (d) 4,4-dimethyl-2-pentyne.arrow_forwardIndicate whether each statement is true or false. (a) Pentanehas a higher molar mass than hexane. (b) The longer the linearalkyl chain for straight-chain hydrocarbons, the higherthe boiling point. (c) The local geometry around the alkynegroup is linear. (d) Propane has two structural isomers.arrow_forwardDraw structures for the following compounds. Show all of the hydrogen atoms in the molecules. (a) ethanol (b) 2-propanol (c) 2-ethyl-5-methylcyclohexanolarrow_forward
- Draw line structures for the following alkenes. Which can exist as cis–trans isomers? For those that can, draw both isomers.(a) 2-Methyloct-2-ene (b) Hept-3-ene(c) 3,4-Dimethylhex-3-enearrow_forward(a) The compound given below had the following IUPAC name and structural formula dibromocyclopentane C3H6CHBrCHBr (i) What type of isomerism is possible in the organic compound? (ii) Draw all the pairs of possible isomers and name them.arrow_forwardWhich compounds exhibit geometric isomerism? Draw and name the two isomers in each case:(a) propene (b) 3-hexene(c) 1,1-dichloroethene (d) 1,2-dichloroethenearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





