
Concept explainers
(a)
Interpretation:
Name of
Concept Introduction:
Electron-dot structure:
This is only way to show the sharing of electrons between atom in polar covalent bonds or covalent bond. This is often called as Lewis structure. This structure denotes valence electrons of an atom by dots and also it shows the way of valence electron distributed within a molecule.
Naming molecular compounds:
To name binary ionic compounds, first predict the positive ion and then negative ion. The positive ion is named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
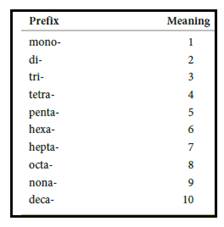
Figure 1
(b)
Interpretation:
Name of
Concept Introduction:
Electron-dot structure:
This is only way to show the sharing of electrons between atom in polar covalent bonds or covalent bond. This is often called as Lewis structure. This structure denotes valence electrons of an atom by dots and also it shows the way of valence electron distributed within a molecule.
Naming molecular compounds:
To name binary ionic compounds, first predict the positive ion and then negative ion. The positive ion is named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
(c)
Interpretation:
Name of
Concept Introduction:
Electron-dot structure:
This is only way to show the sharing of electrons between atom in polar covalent bonds or covalent bond. This is often called as Lewis structure. This structure denotes valence electrons of an atom by dots and also it shows the way of valence electron distributed within a molecule.
Naming molecular compounds:
To name binary ionic compounds, first predict the positive ion and then negative ion. The positive ion is named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
(d)
Interpretation:
Name of
Concept Introduction:
Electron-dot structure:
This is only way to show the sharing of electrons between atom in polar covalent bonds or covalent bond. This is often called as Lewis structure. This structure denotes valence electrons of an atom by dots and also it shows the way of valence electron distributed within a molecule.
Naming molecular compounds:
To name binary ionic compounds, first predict the positive ion and then negative ion. The positive ion is named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
General Chemistry: Atoms First
- Write a Lewis structure for each of the following molecules and ions:(a) (CH3)3SiH(b) SiO44−(c) Si2H6(d) Si(OH)4(e) SiF62−arrow_forwardWrite the chemical formulas for the following compounds:(a) Silver cyanide(b) Calcium hypochlorite(c) Potassium chromate(d) Gallium oxide(e) Potassium superoxide(f) Barium hydrogen carbonatearrow_forwardWhich of the following is NOT a covalent compound? (a) CaCl2 (b) CO2 (c) NO2 (d) CCl4arrow_forward
- Identify the electron pair geometry and the molecular structure of each of the following molecules or ions:(a) IF6+(b) CF4(c) BF3(d) SiF5−(e) BeCl2arrow_forwardWhich of these statements about resonance is true?(a) When you draw resonance structures, it is permissibleto alter the way atoms are connected.(b) The nitrate ion has one long N¬O bond and two shortN¬O bonds.(c) “Resonance” refers to the idea that molecules areresonating rapidly between different bonding patterns.(d) The cyanide ion has only one dominant resonancestructure.(e) All of the above are true.arrow_forwardDraw the Lewis structures and describe the geometry for the following:(a) PF4+(b) PF5(c) PF6−(d) POF3arrow_forward
- Write the Lewis structures for the following, and include resonance structures where appropriate. Indicate which has the strongest carbon-oxygen bond.(a) CO2(b) COarrow_forwardDraw the Lewis structures for CO2 and CO, and predict the number of σ and π bonds for each molecule.(a) CO2(b) COarrow_forwardWrite Lewis structures for the following molecules or ions. (Assign lone pairs, radical electrons, and atomic charges where appropriate.) (a) SbO43− (b) ICl6− (c) SO32- (d) HOBrOarrow_forward
- Which of the following compounds does not contain both ionic and covalent bonding? Justify your answer. (a) Na2SO4(b) NH+NO3(c) NH4C1(d) KCl(e) KclO4arrow_forwardPart A) Out of the following, the species with a single covalent bond is: (a) CO (b) CN- (c) Cl2 (d) N2 Part B) Which of the following compounds has ionic bonds only? (a) NH4ClO4 (b) Mg3N2 (c) Mg3(PO4)2 (c) CHCl3 Part C) The molecular shape and polarity of xenon tetrafluoride are: (a) Tetrahedral & non-polar (b) square planar & non-polar (c) Square pyramidal & polar (d) square planar & polararrow_forwardIdentify the main group to which X belongs in each of the following ionic compound formulas. (a) AlɔX3 (b) X(NO3)2 (c) CaX2arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





