
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10, Problem 10.42SP
Complete the following acid-base reactions. In each case, indicate whether the equilibrium favors the reactants or the products, and explain your reasoning.
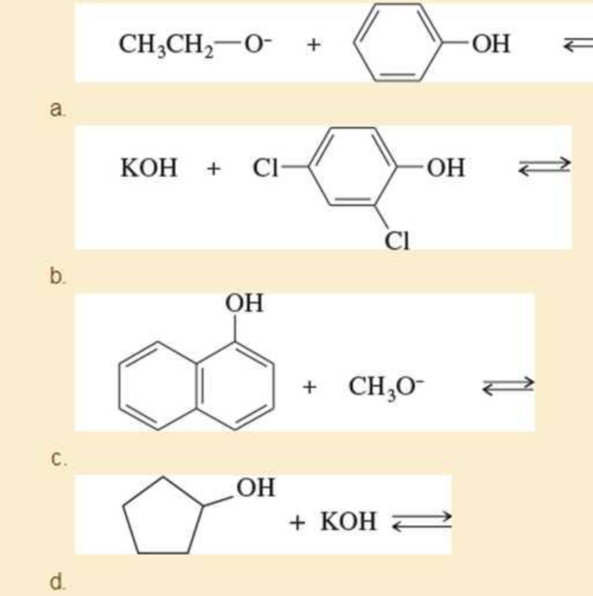
e. (CH3)3C—O− + CH3CH2OH ⇄
f. (CH3)3C—O− + H2O ⇄
g. KOH + CH3CH2OH ⇄
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Rank the following compounds in their correct order of acidity. 1=Most acidic and 4=least acidic.
Rank the following species in order of increasing acidity. Explain your reasons for ordering them as you do. HF NH3 H2SO4 CH3OH CH3COOH H3O+ H2O
When the following reaction reaches equilibrium, which side will the equilibrium lie on- left or right? Please explain in detail.
ClO- + CH3CO2H ⇌ HClO + CH3CO2-
Chapter 10 Solutions
Organic Chemistry (9th Edition)
Ch. 10.3A - Prob. 10.1PCh. 10.3B - Give both the IUPAC name and the common name for...Ch. 10.3B - Prob. 10.3PCh. 10.3C - Give a systematic (IUPAC) name for each diol. a....Ch. 10.4B - Predict which member of each pair will be more...Ch. 10.4B - Dimethylamine (CH3)2NH, has a molecular weight of...Ch. 10.6A - Prob. 10.7PCh. 10.6A - Prob. 10.8PCh. 10.6C - A nitro group (NO2) effectively stabilizes a...Ch. 10.6C - Prob. 10.10P
Ch. 10.8B - Prob. 10.11PCh. 10.8B - Prob. 10.12PCh. 10.9A - Prob. 10.13PCh. 10.9B - Prob. 10.14PCh. 10.9C - Show how you would synthesize each tertiary...Ch. 10.9D - Prob. 10.16PCh. 10.9D - Show how you would add Grignard reagents to acid...Ch. 10.9D - A formate ester, such as ethyl formate, reacts...Ch. 10.9E - Prob. 10.19PCh. 10.9E - In Section9-7B, we saw how acetylide ions add to...Ch. 10.9F - Prob. 10.21PCh. 10.10A - Prob. 10.22PCh. 10.10B - Prob. 10.23PCh. 10.11B - Predict the products you would expect from the...Ch. 10.11B - Prob. 10.25PCh. 10.11B - Prob. 10.26PCh. 10.12 - Prob. 10.27PCh. 10.12 - Prob. 10.28PCh. 10.12 - Authentic skunk spray has become valuable for use...Ch. 10 - Give a systematic (IUPAC) name for each alcohol....Ch. 10 - Give systematic (IUPAC) names for the following...Ch. 10 - Draw the structures of the following compounds...Ch. 10 - Predict which member of each pair has the higher...Ch. 10 - Predict which member of each pair is more acidic,...Ch. 10 - Predict which member of each group is most soluble...Ch. 10 - Draw the organic products you would expect to...Ch. 10 - Prob. 10.37SPCh. 10 - Show how you would synthesize the following...Ch. 10 - Show how you would use Grignard syntheses to...Ch. 10 - Show how you would accomplish the following...Ch. 10 - Show how you would synthesize the following: a....Ch. 10 - Complete the following acid-base reactions. In...Ch. 10 - Prob. 10.43SPCh. 10 - Prob. 10.44SPCh. 10 - Geminal diols, or 1,1-diols, are usually unstable,...Ch. 10 - Vinyl alcohols are generally unstable, quickly...Ch. 10 - Compound A (C7H11Br) is treated with magnesium in...Ch. 10 - Prob. 10.48SPCh. 10 - Prob. 10.49SPCh. 10 - Prob. 10.50SPCh. 10 - Prob. 10.51SPCh. 10 - Prob. 10.52SPCh. 10 - Prob. 10.53SPCh. 10 - Prob. 10.54SPCh. 10 - Prob. 10.55SPCh. 10 - Prob. 10.56SPCh. 10 - Show how this 1 alcohol can be made from the...Ch. 10 - Prob. 10.58SPCh. 10 - Prob. 10.59SPCh. 10 - Prob. 10.60SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach
What is the pH range for acidic solutions? For basic solutions?
EBK INTRODUCTION TO CHEMISTRY
2. Why shouldn’t you work in a laboratory by yourself?
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
141. Design a device that uses as electrochemical cell to determine amount of
in a sample water Describe, in...
Chemistry: Structure and Properties (2nd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Rank the following compounds in order of increasing acidity (1 = least acidic, 3 = most acidic) and in the space provided use resonance (of the conjugate base) to explain why the compound you have labelled “3” is the most acidic.arrow_forward1. Rank the following molecules in terms of increasing acidity. Explain your rankings in terms of stability of the corresponding conjugate base. OH HO, SH NaNH2 =H HO,arrow_forwardDraw an energy diagram for the Brønsted–Lowry acid–base reaction of CH3CO2H with -OC(CH3)3 to form CH3CO2-and (CH3)3COH. Label the axes, starting materials, products, ?H°, and Ea. Draw the structure of the transition state.arrow_forward
- Predict the direction of equilibrium in the following reaction. Explain your answer. 요요 -ll + NH₂ + NH3arrow_forwardhydrogens on a carbon adjacent to a carbonyl group are far more acidic than those not adjacent to a carbonyl group. The anion derived from acetone, for example, is more stable than is the anion derived from ethane. Account for the greater stability of the anion from acetone. CH,ČCH, H CH,CH, H Acetone Ethane pK, 20.2 pK, 51arrow_forwardWhich is a stronger acid? Why? CH3 CO OH or CH3 OSO OHarrow_forward
- Rank the following acids in decreasing acidity. Explain your answer CH2ClCOOH, CHCl2COOH, CH3COOHarrow_forwardState whether the following reactions are favorable or unfavorable. Explain the reason behind your answer.arrow_forwardQ6:- Rank the following substances in order of increasing acidity , Justify your choice. m-bromophenol, m-cresol m-nitrophenol , phenolarrow_forward
- 1. Rank the following species in order of increasing acidity. Explain your reasons for ordering them as you do. HF NH3 H2SO4 CH3OH CH3COOH H3O+ H2O2. Consider the following compounds that vary from nearly nonacidic to strongly acidic. Draw the conjugate bases of these compounds, and explain why the acidity increases so dramatically with substitution by nitro groups. CH4 CH3NO2 CH2(NO2)2 CH(NO2)3arrow_forward4. The following two compounds are protonated when treated with one mole equivalent of a strong acid (e.g. HCI). For each compound, draw the equilibrium to show the preferred site of protonation and give an estimate of the base strength (pKaH). -OHarrow_forwardExplain the trends in the acidity of phenol and the monofluoro derivatives of phenol. OH он он он F pк, 10.0 pK, 8.81 pK, 9.28 pK, 9.81arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY