
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 10.3B, Problem 10.2P
Give both the IUPAC name and the common name for each alcohol.
- a. CH3CH2CH(OH)CH3
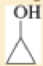
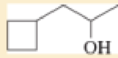
- b. (CH3)2 CHCH2 CH2 OH
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Classify each alcohol as to 1º, 2º, or 3º
1.
2. CH3CH2CH(CH3)CH2OHCH3
3.
CH2CH2CHOHCH3
The correct IUPAC name for this compound is:
O a. 4-oxa-1-cyclohexanol
O b. 4-hydroxyoxacyclohexene
O c. 4-oxa-1-cyclohexenol
O d. 4-oxacyclohexane-1-ol
OH
Which alkyl halide has the highest boiling point? A. CH3BrB. CH3FC. CH3ClD. CH3l
Chapter 10 Solutions
Organic Chemistry (9th Edition)
Ch. 10.3A - Prob. 10.1PCh. 10.3B - Give both the IUPAC name and the common name for...Ch. 10.3B - Prob. 10.3PCh. 10.3C - Give a systematic (IUPAC) name for each diol. a....Ch. 10.4B - Predict which member of each pair will be more...Ch. 10.4B - Dimethylamine (CH3)2NH, has a molecular weight of...Ch. 10.6A - Prob. 10.7PCh. 10.6A - Prob. 10.8PCh. 10.6C - A nitro group (NO2) effectively stabilizes a...Ch. 10.6C - Prob. 10.10P
Ch. 10.8B - Prob. 10.11PCh. 10.8B - Prob. 10.12PCh. 10.9A - Prob. 10.13PCh. 10.9B - Prob. 10.14PCh. 10.9C - Show how you would synthesize each tertiary...Ch. 10.9D - Prob. 10.16PCh. 10.9D - Show how you would add Grignard reagents to acid...Ch. 10.9D - A formate ester, such as ethyl formate, reacts...Ch. 10.9E - Prob. 10.19PCh. 10.9E - In Section9-7B, we saw how acetylide ions add to...Ch. 10.9F - Prob. 10.21PCh. 10.10A - Prob. 10.22PCh. 10.10B - Prob. 10.23PCh. 10.11B - Predict the products you would expect from the...Ch. 10.11B - Prob. 10.25PCh. 10.11B - Prob. 10.26PCh. 10.12 - Prob. 10.27PCh. 10.12 - Prob. 10.28PCh. 10.12 - Authentic skunk spray has become valuable for use...Ch. 10 - Give a systematic (IUPAC) name for each alcohol....Ch. 10 - Give systematic (IUPAC) names for the following...Ch. 10 - Draw the structures of the following compounds...Ch. 10 - Predict which member of each pair has the higher...Ch. 10 - Predict which member of each pair is more acidic,...Ch. 10 - Predict which member of each group is most soluble...Ch. 10 - Draw the organic products you would expect to...Ch. 10 - Prob. 10.37SPCh. 10 - Show how you would synthesize the following...Ch. 10 - Show how you would use Grignard syntheses to...Ch. 10 - Show how you would accomplish the following...Ch. 10 - Show how you would synthesize the following: a....Ch. 10 - Complete the following acid-base reactions. In...Ch. 10 - Prob. 10.43SPCh. 10 - Prob. 10.44SPCh. 10 - Geminal diols, or 1,1-diols, are usually unstable,...Ch. 10 - Vinyl alcohols are generally unstable, quickly...Ch. 10 - Compound A (C7H11Br) is treated with magnesium in...Ch. 10 - Prob. 10.48SPCh. 10 - Prob. 10.49SPCh. 10 - Prob. 10.50SPCh. 10 - Prob. 10.51SPCh. 10 - Prob. 10.52SPCh. 10 - Prob. 10.53SPCh. 10 - Prob. 10.54SPCh. 10 - Prob. 10.55SPCh. 10 - Prob. 10.56SPCh. 10 - Show how this 1 alcohol can be made from the...Ch. 10 - Prob. 10.58SPCh. 10 - Prob. 10.59SPCh. 10 - Prob. 10.60SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
45. Calculate the mass of nitrogen dissolved at room temperature in an 80.0-L home aquarium. Assume a total pre...
Chemistry: Structure and Properties
Characterize each of the following structures as aromatic, nonaromatic, or antiaromatic:
Answer: _____
Organic Chemistry As a Second Language: Second Semester Topics
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living By Chemistry: First Edition Textbook
Describe the orbitals used in bonding and the bond angles in the following compounds: a. CH3O b. CO2 c. H2CO d....
Organic Chemistry (8th Edition)
Write a Lewis formula for each of the following organic molecules: C2H3Cl (vinyl chloride: starting material fo...
Organic Chemistry - Standalone book
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Name each compound in which the benzene ring is best treated as a substituent. CH3 a. CH3-CH-CH,-CH-CH,-CH–CH,-CH; CH,-CH3 b. CH,-CH-CH=CH-CH,-CH,–CH,-CH, c. CH3-C=C-CH-CH-CH-CH2-CH3 CH3 CH3arrow_forward16. Identify each compound as an a cohol, a phenol, or an ether. Classify any alcohols as primary (1"), secondary (2), or tertiary (3"). a. CH,CH,CH,OH CH,CHCH, b. CHO C. CH CHOCH, CH, d.arrow_forwardList the products of each alcohol reaction. CH3 a. CH,-C-OH CH, NazCrO b. CH3-CH-CH;-CH2-OH c. CH-CH-OH +HCI -arrow_forward
- 18. Ketone reduction Dicyclohexyl ketone Reduce the ketone. 1. NaBH4, ethanol 2. H3O+ H OH Dicyclohexylmethanol (88%) (a 2° alcohol)arrow_forwardWhat is the correct IUPAC name for the following compound? CH3- CH- CH2-CH- CH- CH3 CH3 a. 5-methyl-2-hexanol O b. 2,5-dimethyl-5-pentanol O c. 1,4-dimethyl-1-pentanol O d. 5,5-dimethyl-2-pentanol O e. 2-methyl-5-hexanol OHarrow_forwardDraw the products of combustion of each alkane. a. CH;CH,CH,CH2CH(CH3)2 b.arrow_forward
- Classify each alkyl halide as 1°, 2°, or 3°. CH3 c. CHg-C-CHCH3 ČH3 ČI CH;CH2CH,CH,CH2-Br b. d. a.arrow_forwardB. Give the IUPAC name for each compound. F CH3 ÇI 1. CH;CH2CHCH2CH2CHCH3 5. CH,CH3 F 2. CI 6. OH CI NO, 7. NO, NO, Br 8. Br 3.arrow_forwardList the products of each alcohol reaction. a. CH3-CH2-CH,-OH +HBr - H,S0, b. CH3-CH-CH2-OH с. СНу— CH—он +Na CH3 Na:Cr0 d. CH3-C-CH;-CH;-OH CH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY