
Interpretation:
The equation for the reaction between acetic acid and
Concept introduction:
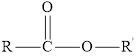
Here, R and
The steps to write the IUPAC name of an ester are as follows:
Step 1: Identify the parent chain. The alkyl group from the parent acid represents the parent chain.
Step 2: Number the carbon atoms. Since the parent chain is derived from carboxylic acid, therefore; the name of the main acid is alkanoic acid. Drop the letters
Step 3: Number the carbon atoms of the alcohol part from the carbon which is directly bonded to the oxygen atom. The alcohol part is named as ‘alkyl’.
Step 4: In the case of ester, an alkyl group of the alcohol is written first followed by the anion of carboxylic acid. The format to name an ester is as follows:
Trending nowThis is a popular solution!

Chapter 21 Solutions
Introductory Chemistry: An Active Learning Approach
- Write the two step reaction that would produce benzoic acid from benzenearrow_forwardWrite three difference between ethanol and ethanoic acid on the basis of chemical properties?arrow_forwardWhat are the generic structures for carboxylic acids and esters? Write a structure for a specific carboxylic acid and ester.arrow_forward
- How do carboxylic acids and esters differ in their characteristic? Why are there differences in the solubility of the two acids tested? Write the complete (including states) balanced chemical reaction that occurs when acetic acid reacts with sodium bicarbonate.arrow_forwardDraw and Name the condensed structures of the compounds formed from benzoic acid and 2-propanol.arrow_forwardWhat chemical test will you use to differentiate methyl salicylate from methanol?arrow_forward
- When methanoic acid reacts with a mixture of these two propanol isomers, two esters are formed. Draw the complete structural formulas of these two esters.arrow_forwardAldehydes and ketones can be named in a systematic way by counting the number of carbon atoms (including the carbonyl carbon) that they contain. The name of the aldehyde or ketone is based on the hydrocarbon with the same number of carbon atoms. The ending -al for aldehyde or -one for ketone is added as appropriate. Draw the structural formulas for propanal.arrow_forwardExplain the process of Naming Carboxylic Acids ?arrow_forward
- Give a chemical test to distinguish between 2-Pentanol and 3-Pentanol.arrow_forwardDefine the structure of a carboxylic acid ?arrow_forwardBriefly explain why alcoholic solutions (alcoholic NH2OHꞏHCl, alcoholic KOH, alcoholic HCl) are used in the hydroxamic acid test for esters, what happens if we use other solutions?arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning



