
Concept explainers
Interpretation:
Lewis structure of
Concept introduction:
The structure of ammonia is,
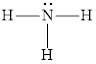
The nitrogen-containing organic compounds are known as amines. They are basic in nature and lone pairs of electrons are present on the nitrogen atom. They can be called as derivatives of ammonia. Alkyl group substitution for the hydrogen atoms in the ammonia molecule forms an amine.
Amines are named by the alkyl groups first in the order of alphabetical order followed by the suffix amine. When two or more than two same alkyl groups are present, then they are named as di-, tri-, tetra- and so on.
Lewis diagram is the representation of the molecule or ion. It shows the bonding between the atoms and also shows the unshared pairs of electrons on an atom.
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
Introductory Chemistry: An Active Learning Approach
- Name an amine with 7 carbonsarrow_forwardTRUE OR FALSE (a) There are three amines with the molecular formula C3H9N. (b) Aldehydes, ketones, carboxylic acids, and esters all contain a carbonyl group. (c) A compound with the molecular formula of C3H6O may be either an aldehyde, a ketone, or a carboxylic acid. (d) Bond angles about the carbonyl carbon of an aldehyde, a ketone, a carboxylic acid, and an ester are all approximately 109.5°. (e) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O. (f) The molecular formula of the smallest carboxylic acid is C2H4O2.arrow_forwardWrite the systematic (IUPAC) names for the amines. The names should have the format alkanamine. H₂C-N-CH3 H3C-CH2-CH-CH3 systematic (IUPAC) name: HC-N-CH2-CH3 HC-CH2-CH2-CH₂ systematic (IUPAC) name: These compounds are amines.arrow_forward
- Draw the skeletal ("line") structure of a secondary amine with 4 carbon atoms, and no double or triple bonds.arrow_forwardWrite the common name for each amine. HỌC—N—CH,CH3 H₂C-CH₂-CH₂-CH₂ Ethylbutylamine common name: Incorrect HC—CH,—N—CH,—CHy CH₂ CH3 diethylethylamine common name: Incorrect These compounds are tertiary amines.arrow_forwardThree amide isomers, N,N-dimethylformamide, N-methylacetamide, and propanamide, have respective boiling points of 153 °C (426 K), 202 °C (475 K), and 213 °C (486 K). Explain these boiling points in light of their structural formulas.arrow_forward
- Write the names for the amines using the naming styles taught in the McMurry text. H2 H3C-N-C-CH3 name: H2 H2 H3C-C-Ņ-c-CH3 H name: These compounds are amines.arrow_forwardWhen amines are reacted with bases, they form ammonium salts. Is that true or false?arrow_forwardExplain aryl and alkyl amines and their reactions by giving two examples of each.arrow_forward
- Describe the water solubility of amines in relation to theircarbon chain length.arrow_forwardBriefly explain primary, secondary, tertiary, and quarternary amine structures.arrow_forwardDraw structural formulas for all the amines having the formula C3H9N. Classify each as either a primary amine, a secondary amine, or a tertiary amine.arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
