
(a)
Interpretation:
From the given pictures, the material that relates to it has to be identified.
Concept introduction:
Semiconductor: A material which can conduct electricity in range between a metal and an insulator is known as semiconductor. Addition of small amount of impurities increases conductivity of semiconductor and the process is known as doping. n-type and p-type are two types of doped semiconductors.
n-type: This is a type of doped semiconductor in which many free electrons are present in conduction band.
p-type: This is a type of doped semiconductor in which holes are present in conduction band.
Insulator: Insulators are one, in which electrons are filled in valence band with a vacant conduction band and they are parted away by band gap.
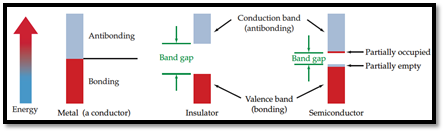
Figure 1
(b)
Interpretation:
The ascending order of electrical conductivity of given materials has to be arranged.
Concept introduction:
Semiconductor: A material which can conduct electricity in range between a metal and an insulator is known as semiconductor. Addition of small amount of impurities increases conductivity of semiconductor and the process is known as doping. n-type and p-type are two types of doped semiconductors.
n-type: This is a type of doped semiconductor in which many free electrons are present in conduction band.
p-type: This is a type of doped semiconductor in which holes are present in conduction band.
Insulator: Insulators in which electrons are filled in valence band with a vacant conduction band and they are parted away by band gap. Insulators does not conduct electricity.
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
General Chemistry: Atoms First
- 8.96 A business manager wants to provide a wider range of p- and n-type semiconductors as a strategy to enhance sales. You are the lead materials engineer assigned to communicate with this manager. How would you explain why there are more ways to build a p-type semiconductor from silicon than there are ways to build an n-type semiconductor from silicon?arrow_forwardCarbon exists as several allotropes including buckminsterfullerene and carbonnanotubes. All are composed of pure carbon, but their materials properties differdramatically due to the different arrangements of atoms in their structures. Name TWO(2) other allotropes and compare both of them in terms of:i) crystal structure with a suitable diagram.ii) electrical propertiesarrow_forwardwhat is the difference between ferromagnetic and paramagnetic?arrow_forward
- Is Aluminium is commonly deliberately alloyed with iron? Why ?arrow_forwardWhich groups of elements have the best and which the poorest electrical conductivities? Explain.arrow_forwardIndicate which of the following statements are true with regard to semiconductors. (a) elemental semiconductors are made of metalloids (b) semiconductors have a band gap that is greater than 3.5 eV (c) doping a semiconductor with a few ppm of a p block element increases the conductivity of that material by a million fold (d) compound semiconductors must contain at least one metalloid elementarrow_forward
- The density of lead is 11.35 g cm-3 and the metal crystallizes with fee unit cell. Estimate the radius of lead atom. (At Mass of lead = 207 g mol-1 and NA = 6.02 X 1023mol-1arrow_forwardPhosphorus (P) is present in a germanium (Ge) sample. Assume that one of its five valence electrons revolves in a Bohr orbit around each P+ ion in the Ge lattice. (a) If the effective mass of the electron is 0.17 me and the dielectric constant of Ge is 16, find the radius of the first Bohr orbit of the electron. (b) Ge has a band gap (Eg) of 0.65 eV. How does the ionization energy of the above electron comparing to Eg and kBT at room temperature?arrow_forwardSilicon carbide, SiC, has the three-dimensional structure shown in the figure. (a) Name another compound that has the same structure.(b) Would you expect the bonding in SiC to be predominantlyionic, metallic, or covalent? (c) How do the bondingand structure of SiC lead to its high thermal stability(to 2700 °C) and exceptional hardness?arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning


