
Concept explainers
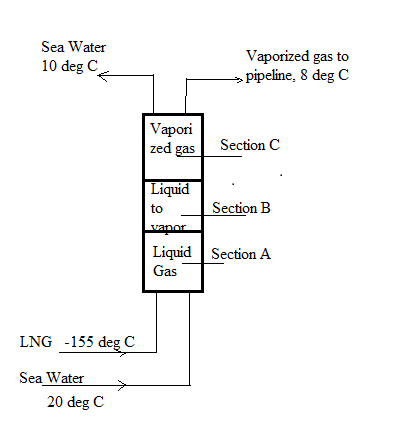
A liquefied natural gas (LNG) regasification facilityutilizes a vertical heat exchanger or vaporizer that consists of a shell with a single-pass tube bundle used toconvert the fuel to its vapor form for subsequent delivery through a land-based pipeline. Pressurized LNG isoff-loaded from an oceangoing tanker to the bottom ofthe vaporizer at
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Fundamentals of Heat and Mass Transfer
Additional Engineering Textbook Solutions
Introduction To Finite Element Analysis And Design
Fox and McDonald's Introduction to Fluid Mechanics
Fundamentals Of Thermodynamics
Engineering Mechanics: Statics
Degarmo's Materials And Processes In Manufacturing
Thinking Like an Engineer: An Active Learning Approach (3rd Edition)
- 1. A. Using the Steam Tables, determine the amount of cooling water needed by a condenser in kg/sec if wet steam comes into the condenser at P= 610.44mmHG vac and 99.47 % moisture, mass flow rate= 7200+ (X/SN) kg/hr. Cooling water enters the condenser at 12 deg C and leaves at 21 deg C. Answer: B. Draw the schematic diagram of the condenser and process in the PV and TS diagrams below. Cp H20 = 4.187 KJ/(kg-K): Schematic diagram of heat exchanger =condenser S 2. Use only the MD below to solve this Item; use it by plotting data and process to determine items being asked. Steam enters a turbine at 117.4308 atmgage and 932 deg. F, and expands isentropicaly to 177.17 inches of Hg. For 2 + (X/SN) kg/sec of steam, what is (a) the ideal work done (KI/sec) if AKE = 0 and actual exhaust enthalpy is 2700 KI/kg. a)Ans: (b)ne =_ (c)Determine: (e) Actual steam temp ideal steam moisture at exhaust. Ans: at the exhaust. Ans. g) change in entropy during the actual process. Ans 3000 2500 2000…arrow_forwardA heat exchanger, shown below, is used to cool an air flow from 550 K to 250 K, both states at 1 MPa. The coolant is a water flow at 30 °C, 0.1 MPa. If the water leaves as saturated vapor, 0.1 MPa. 1 air 3 water The initial state enthalpy for air (h;(kJ/kg)) is equal to: The final state enthalpy for air (h2(kJ/kg)) is equal to: The initial state enthalpy for water (h3(kJ/kg)) is equal to: The final state enthalpy for water (ha(kJ/kg)) is equal to: Mµ20/Mair| The ratio of the flow ratesarrow_forwardE6 Air handling supplies 50 m^3/s of air mixed by 20% by weight of fresh air at 45 C (DBT) and 30 C(WBT) with 80% of recirculated air from the space at 25 C(DBT) and 50% RH. The air leaves the cooling coil at 12 C saturated state. The cooling capacity and the moisture removal rate from the supplied air to * :the space are cooling coil 120 Saturation Fresh air A room 45C(DBT) 30c(WBT) m=50 m^3/s r 250 RH=50% exhaust air r 996.82 kw, 0.0532 kg/s O 1480.65 kW, 0.1875 kg/s O 536.17 kw, 0.0174 kg/s O 1731.05 kW, 0.216 kg/s O 510.92 kW, 0.0153 kg/sarrow_forward
- Q4//A// Prove for the condenser: + %3D Cpw (Twz-Tw1) when the temperature of cooling water outlet and inlet is equal to saturation temperature of steam and the condensate leaving the condenser respectively; also, the inlet of condenser is dry steam with unity latent heat. m, Twiarrow_forwardQuestion (1) 1. What are the different methods of the water treatments for boiler make-up water? 2. Determine the power required for a forced draught and induced draught fan in a boiler having coal consumption of 25 komin and airlfuel ratio of 19 by mass. The ambient, air temperature and hot gas temperature at exit are 30 °C and 140 °C respectively. Draught required is equivalent to 32.2 mm of water and mechanical efficiency of motor/fan is merely 70%arrow_forwardThe following properties for Freon-12 were obtained. At -22 °C h (superheat) = 353 KJ/kg ha = 343 KJ/kg %3D At 44 °C h (superheat) = 300 KJ/kg h (subcooled) = 228 KJ/kg h = 238 KJ/kg %3D %3D Vapor Compression Refrigeration system with Heat Exchanger uses Freon-12. Liquid refrigerant leaves the condenser at 44 °C. The evaporator Temperature is -22°C. The internal heat exchanger temperature is 30°C. Compute for the cooling effect in KJ/kg.arrow_forward
- What is the ideal mass flow rate for a refrigeration and cryogenic plant for oxygen production? Show solution if necessary and reference.arrow_forward1. A. Using the Steam Tables, determine the amount of cooling water needed by a condenser in kg/sec if wet steam comes into the condenser at P= 610.44mmHG vac and 99.47 % moisture , mass flow rate = 7200 + (SN/X) kg/hr. Cooling water enters the condenser at 12 deg C and leaves at 21 deg C. Answer: B. Draw the schematic diagram of the condenser and process in the PV ahd TS diagrams below. Cp H2O = 4.187 KJ/(kg-K): Schematic diagram of heat exchanger =condenserarrow_forwardWater is used in a heat exchanger to cool 5 kg/s of air from 400 to 200 C. Calculate the minimum mass flow rate of the water if Twater =10 C.arrow_forward
- 1. A. Using the Steam Tables, determine the amount of cooling water needed by a condenser in kg/sec if wet steam comes into the condenser at P= 610.44mmHG vac and 99.47 % moisture, mass flow rate = 7200 + (10/13) kg/hr. Cooling water enters the condenser at 12 deg Cand leaves at 21 deg C. Answer: diagrams below. Cp H20 = 4.187 KJ/kg-K): Schematic diagram of heat exchanger =condenser B. Draw the schematic diagram of the condenser and process in the PV and TS V 2. Use only the MD below to solve this Item; use it by plotting data and process to determine items being asked. Steam enters a turbine at 117.4308 atmgage and 932 deg. F, and expands isentropicaly to 177.17 inches of Hg. For 2 + (10/13 kg/sec of steam, what is (a) the ideal work done (KJ/sec) if AKE = 0 and actual exhaust enthalpy is 2700 KJ/kg. a)Ans: (b)ne =. (c]Determine: ideal steam moisture at exhaust. Ans: (d) Actual steam quality at exhaust Ans: (e) Actual steam temp g) change in entropy during the actual process. at the…arrow_forwardA simple air cooled system is used for an aero plane to take a load of 10 tons.Atmospheric temperature and pressure is 25°C and 0.9 atm respectively. Dueto ramming the pressure of air is increased from 0.9 atm, to 1 atm. The pressureof air leaving the main compressor is 3.5 atm and its 50% heat is removed inthe air-cooled heat exchanger and then it is passed through a evaporator for future cooling. The temperature of air is reduced by 10°C in the evaporator.Lastly the air is passed through cooling turbine and is supplied to the coolingcabin where the pressure is 1.03 atm. Assuming isentropic efficiency of the compressor and turbine are 75% and 70%,find i) Power required to take the load in the cooling cabinii) COP of the system.The temperature of air leaving the cabin should not exceed 25°Carrow_forwardA one-effect evaporator operates with a solution whose properties are similar. to pure water. The feed flow is 60000 kg/h, with a concentration of 20% (in mass) and temperature of 30 °C. Knowing that the concentration at the exit is 40% (in mass) and that the operating pressure of the barometric condenser is 0.075 bar, estimate the consumption of saturated steam in kg/s at 2 bar.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





