
Concept explainers
(a)
Interpretation: The structure of the given coordination complex has to be drawn. The oxidation state, coordination number and the coordination geometry of the metal should be determined.
Concept introduction:
Coordination compounds are a special class of compounds in which the metal atoms or ions are bounded to a number of anions or neutral molecules.
A coordination compound can be simply represented as follows,
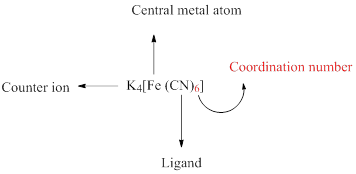
Oxidation number is the charge of the central atom of a coordination complex and it is represented by roman numerals.
Coordination number of a complex is defined as the number of ligand donor atoms to which the metal is directly bonded.
The geometry of a
Coordination number of complexes, shape with example are listed below,
(b)
Interpretation: The structure of the given coordination complex has to be drawn. The oxidation state, coordination number and the coordination geometry of the metal should be determined.
Concept introduction:
Coordination compounds are a special class of compounds in which the metal atoms or ions are bounded to a number of anions or neutral molecules.
A coordination compound can be simply represented as follows,

Coordination number of a complex is defined as the number of ligand donor atoms to which the metal is directly bonded.
Oxidation number is the charge of the central atom of a coordination complex and it is represented by roman numerals. The geometry of a transition metal complex depends on the coordination number and nature of the metal ion.
Coordination number of complexes, shape with example are listed below,
(c)
Interpretation: The structure of the given coordination complex has to be drawn. The oxidation state, coordination number and the coordination geometry of the metal should be determined.
Concept introduction:
Coordination compounds are a special class of compounds in which the metal atoms or ions are bounded to a number of anions or neutral molecules.
A coordination compound can be simply represented as follows,

Coordination number of a complex is defined as the number of ligand donor atoms to which the metal is directly bonded.
Oxidation number is the charge of the central atom of a coordination complex and it is represented by roman numerals.
The geometry of a transition metal complex depends on the coordination number and nature of the metal ion.
Coordination number of complexes, shape with example are listed below,
(d)
Interpretation: The structure of the given coordination complex has to be drawn. The oxidation state, coordination number and the coordination geometry of the metal should be determined.
Concept introduction:
Coordination compounds are a special class of compounds in which the metal atoms or ions are bounded to a number of anions or neutral molecules.
A coordination compound can be simply represented as follows,

Coordination number of a complex is defined as the number of ligand donor atoms to which the metal is directly bonded.
Oxidation number is the charge of the central atom of a coordination complex and it is represented by roman numerals.
The geometry of a transition metal complex depends on the coordination number and nature of the metal ion.
Coordination number of complexes, shape with example are listed below,
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
General Chemistry: Atoms First
- What is the coordination number of the central metal atom in the following complexes? (a) [Fe(H2O)63+] (b) [Pt(NH3)Br3] (c) [V(en)Cl42] (d) [Au(CN)2+]arrow_forwardPlatinum(II) forms many complexes, among them those with the following ligands. Give the formula and charge of each complex. (a) two ammonia molecules and one oxalate ion (C2O42-) (b) two ammonia molecules, one thiocyanate ion (SCN-), and one bromide ion (c) one ethylenediamine molecule and two nitrite ionsarrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





