
Concept explainers
(a)
Interpretation:
The products of the proton transfer reaction shown below are to be drawn.
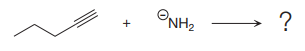
Concept introduction:
In proton transfer reaction, a Bronsted-Lowry base reacts with a Bronsted-Lowry acid. The Bronsted-Lowry acid donates a proton and forms a conjugate base while the Bronsted-Lowry base accepts a proton and forms a conjugate acid. The proton transfer reaction consists of a single elementary step.
(b)
Interpretation:
A free energy diagram for the following reaction, indicating whether it is endothermic or exothermic, is to be drawn.
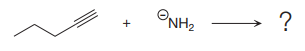
Concept introduction:
When comparing two acids, the acid with the lower
A reaction tends to be spontaneous if
Trending nowThis is a popular solution!

Chapter 6 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Two possible proton transfer reactions can take place ОН + → ? between the reactants shown here. (a) Write the products of each possible proton transfer reaction. (b) Determine which reaction is more energetically favorable.arrow_forwarda.) Rank the attached compounds in order of increasing acidity. (b)Which compound forms the strongest conjugate base?arrow_forwardCan you help me draw the mechanism arrows for this reaction and determine if the reaction is going forward or reverse?arrow_forward
- 2. The following pair of compounds can undergo an acid/base reaction. a) Identify the base and the acid b) Draw the products of the reaction and a mechanism for the proton transfer. c) Based on qualitative concepts, is this reaction favored? Explain. Siarrow_forwardBased on the diagram and given reaction, Which of the compound will be placed on the positive and negative terminal? Explain briefly the reasonarrow_forwardGiven the curved arrow notation for each of the following proton transfer reactions, draw the appropriate products. (a) (b) ? OH: (c) (d) H- ? ? :0arrow_forward
- Draw the mechanism for the proton transfer step. Explain why the products of this step are favored over the reactants.arrow_forward(b) Addition of HBr to the alkene in the presence of peroxide form radical intermediates. Based on the following reaction, use the curved arrows to HBr B (preferred product) ROOR (i) show the initiation step of this reaction. (ii) draw the possible structures of the two radicals formed in the propagation step. (ii) explain the formation of the preferred product B.arrow_forwardDraw the mechanism arrows for the reaction between an acid chloride and an alcohol.arrow_forward
- Draw the arrows for the reaction occuring.arrow_forwarda) Draw the product(s) of the acid/base reaction. b) Will the equilibrium lie left or right?arrow_forwardI understand where the proton transfer occurs but not what the products will look like. Can you draw curved arrows to show proton transfer and what the reaction products would look like? And explain which side is favored and whyarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
