
Concept explainers
(a)
Interpretation:
A reactant W can undergo two separate reactions to yield either X or Y. From the free energy diagram given below, the product that would be in greater abundance at equilibrium is to be determined.
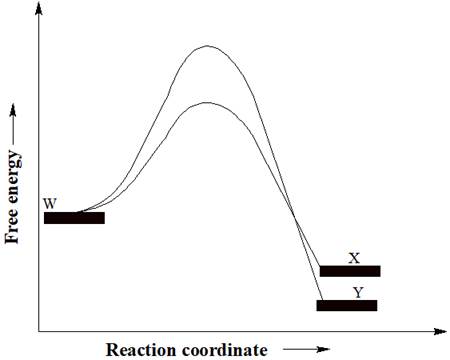
Concept introduction:
In a reaction free energy diagram, Gibbs free energy is plotted as a function of the reaction coordinate. As the reaction coordinate increases, the geometries of the species involved in the reaction increasingly resemble those of the products. The product having lower energy signifies that it is the most stable product. The most stable product would be in greater abundance at equilibrium.
(b)
Interpretation:
A reactant W can undergo two separate reactions to yield either X or Y. From the free energy diagram given below, the product that would be in greater abundance at equilibrium is to be determined.
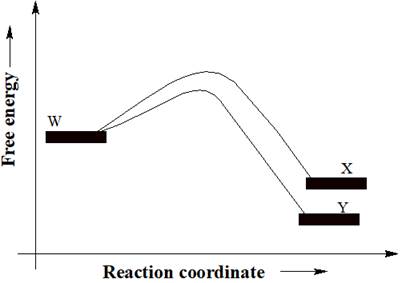
Concept introduction:
In a reaction free energy diagram, Gibbs free energy is plotted as a function of the reaction coordinate. As the reaction coordinate increases, the geometries of the species, involved in the reaction, increasingly resemble those of the products. The product having lower energy signifies that it is the most stable product. The most stable product would be in greater abundance at equilibrium.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- The conversion of reactant R to product A is under control and is preferred at temperature kinetic, low kinetic, high thermodynamic, low thermodynamic, high The conversion of reactant R to product B is under control and is preferred at temperature kinetic, low kinetic, high thermodynamic, low thermodynamic, higharrow_forwardEnergy Type here to search Which of the following statements is true regarding product 1 and 2? Product 1 $ - Product 2 Reaction Coordinate Question 15 of 27 % & A) Product 1 is the kinetic product. Product 2 is the thermodynamic product. B) Product 1 is the thermodynamic product. Product 2 is the kinetic product. C) Product 1 is both the kinetic and thermodynamic product. D) Product 2 is both the kinetic and thermodynamic product. PrtScr F12 Home ENG Su 5:33 PM 7/22/2023arrow_forwardSelect the reactions in Figure 39 that can be represented by the accompanying energy diagram?arrow_forward
- Which side of this equilibrium is favored? Reactants because they are more stable Reactants because they are less stable Products because they are more stable Products because they are less stablearrow_forwardCheck off boxes if they react. Then explain reaction for each one and rank from most to least reactivearrow_forwardWhich of the reactions has a product that is thermodynamically stable compared with the reactant?arrow_forward
- 1. Addition of HBr to alkenes occurs according to rule. Your answerarrow_forwardThe following is generic depiction of a reaction using the curve arrow formalism. -D Which of these statements is not correct for this reaction? Electrons move from C to B. Electrons move from B to A. O In the products, a bond forms between C and B. O In the products, A would have a positive charge.arrow_forwardMatch the following reactions with their reaction type.arrow_forward
- organic chemistry When learning about substitution or elimination reaction, hydrogen or carbon bonding in intimidate move to carbocation, making carbocation more substituted and stable. In here, I can't recall what this situation is calledarrow_forwardDraw the organic products M and N and classify the chemical transformations reactionsarrow_forwardWhat is the effect on the rate constant and reaction rate when reaction temperature is increased? The rate constant will be greater, and the reaction rate will be higher. The rate constant will be smaller, but the reaction rate will be higher. The rate constant will be smaller, and the reaction rate will be lower.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





