
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7.SE, Problem 29MP
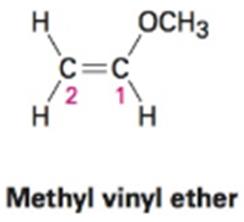
When methyl vinyl ether reacts with a strong acid, the proton adds to C2 exclusively, instead of C1 or the oxygen atom. Draw the three protonated forms of methyl vinyl ether and explain this observation.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
The compound below is treated with chlorine in the presence of light.
H₂C
CH3
H3C
CH₂CH3
Draw the structure for the organic radical species produced by reaction of the compound with a chlorine
atom. Assume reaction occurs at the weakest C-H bond.
• You do not have to consider stereochemistry.
• You do not have to explicitly draw H atoms.
Reaction of (CH3)3CH with Cl2 forms two products: (CH3)2CHCH2Cl (63%) and (CH3)3CCl (37%). Why is the major product formed by cleavage of the stronger 1° C–H bond?
Break compound A into several small (less or equals to 6 carbon atom) molecules. You need to explain the reasons why you choose to break those bonds.
Chapter 7 Solutions
Organic Chemistry
Ch. 7.2 - Calculate the degree of unsaturation in each of...Ch. 7.2 - Prob. 2PCh. 7.2 - Diazepam, marketed as an antianxiety medication...Ch. 7.3 - Give IUPAC names for the following compounds:Ch. 7.3 - Prob. 5PCh. 7.3 - Prob. 6PCh. 7.3 - Prob. 7PCh. 7.4 - Prob. 8PCh. 7.4 - Prob. 9PCh. 7.4 - Prob. 10P
Ch. 7.5 - Which member in each of the following sets ranks...Ch. 7.5 - Prob. 12PCh. 7.5 - Prob. 13PCh. 7.5 - Prob. 14PCh. 7.6 - Prob. 15PCh. 7.8 - Prob. 16PCh. 7.8 - Prob. 17PCh. 7.9 - Show the structures of the carbocation...Ch. 7.9 - Draw a skeletal structure of the following...Ch. 7.10 - Prob. 20PCh. 7.11 - On treatment with HBr, vinylcyclohexane undergoes...Ch. 7.SE - Prob. 22VCCh. 7.SE - Prob. 23VCCh. 7.SE - The following carbocation is an intermediate in...Ch. 7.SE - Prob. 25VCCh. 7.SE - Predict the major product and show the complete...Ch. 7.SE - Prob. 27MPCh. 7.SE - When 1, 3-butadiene reacts with one mole of HBr,...Ch. 7.SE - When methyl vinyl ether reacts with a strong acid,...Ch. 7.SE - Addition of HCl to 1-isopropylcyclohexene yields a...Ch. 7.SE - Addition of HCl to...Ch. 7.SE - Limonene, a fragrant hydrocarbon found in lemons...Ch. 7.SE - Prob. 33MPCh. 7.SE - Calculate the degree of unsaturation in the...Ch. 7.SE - Prob. 35APCh. 7.SE - Prob. 36APCh. 7.SE - Name the following alkenes:Ch. 7.SE - Draw structures corresponding to the following...Ch. 7.SE - Prob. 39APCh. 7.SE - Prob. 40APCh. 7.SE - Prob. 41APCh. 7.SE - Prob. 42APCh. 7.SE - Prob. 43APCh. 7.SE - Draw and name the 17 alkene isomers, C6H12,...Ch. 7.SE - Prob. 45APCh. 7.SE - Prob. 46APCh. 7.SE - Which of the following E, Z designations are...Ch. 7.SE - Prob. 48APCh. 7.SE - trans-2-Butene is more stable than cis-2-butene by...Ch. 7.SE - Prob. 50APCh. 7.SE - Normally, a trans alkene is more stable than its...Ch. 7.SE - trans-Cyclooctene is less stable than...Ch. 7.SE - Prob. 53APCh. 7.SE - Prob. 54APCh. 7.SE - Use Hammond’s Postulate to determine which...Ch. 7.SE - Prob. 56APCh. 7.SE - Predict the major product in each of the following...Ch. 7.SE - Prob. 58APCh. 7.SE - Prob. 59APCh. 7.SE - Prob. 60APCh. 7.SE - Allene (1,2-propadiene), H2C=C=CH2, has two...Ch. 7.SE - The heat of hydrogenation for allene (Problem...Ch. 7.SE - Retin A, or retinoic acid, is a medication...Ch. 7.SE - Prob. 64APCh. 7.SE - tert-Butyl esters [RC02C(CH3)3] are converted into...Ch. 7.SE - Vinylcyclopropane reacts with HBr to yield a...Ch. 7.SE - Prob. 67APCh. 7.SE - Prob. 68APCh. 7.SE - Prob. 69APCh. 7.SE - Prob. 70APCh. 7.SE - Prob. 71APCh. 7.SE - Reaction of 2, 3-dimethyl-l-butene with HBr leads...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- When 2-chloropropane treated with NaOH and 1-chloropropane treated with NaOH separately produce two different functional groups. Provide both reactions and explain the two different functional groups produced.arrow_forwardWhen bromine is added to two beakers, one containing phenyl isopropyl ether and the other containing cyclohexene, the bromine color in both beakers disappears. What observation could you make while performing this test that would allow you to distinguish the alkene from the aryl ether?arrow_forwardGive a test to distinguish between propan-2-one and pentan-3-one.arrow_forward
- Define Electrophilic addition and the Hammond postulate ?arrow_forward5. Isomeric compounds that rapidly interconvert by the movement of a proton like for example ketone formation from enol.arrow_forwardKetones and aldehydes are hydrated under acidic or basic conditions. An example of the acid-catalyzed hydration is shown. Complete the first step of this mechanism. Include all lone pairs of electrons, curved arrows, and nonzero formal charges in your mechanism.arrow_forward
- Linolenic acid (Table 10.2) and stearidonic acid are omega-3 fatty acids, unsaturated fatty acids that contain the first double bond located at C3, when numbering begins at the methyl end of the chain. Predict how the melting point of stearidonic acid compares with the melting points of linolenic and stearic acids. A current avenue of research is examining the use of soybean oil enriched in stearidonic acid as a healthier alternative to vegetable oils that contain fewer degrees of unsaturation.arrow_forwardCompounds A, B and C are chiral and optically active. Determine if the reaction conditions below will cause the ketones to lose their optical activity. Provide an explanation for which compounds will no longer be optically active and which will maintain their optical activity.arrow_forwardRank the following compound in terms of increasing predicted boiling points. Explain your answer. Compounds: Benzene Ring, phenol, dichlorobenzene, Phenylarrow_forward
- Draw a curved arrow mechanism to show the two resonance forms of the intermediate formed in the second electrophilic addition steps. You will need to modify the structure of the starting material to show this intermediate. Remember to include lone pairs and all nonzero formal charges. الملاحة Draw the structure of the intermediate in the second electrophilic addition and curved arrows to show resonance.arrow_forwardAcid-catalyzed dehydration of 3,3-dimethyl-2-butanol gives three alkenes: 2,3-dimethyl-2-butene, 3,3-dimethyl- 1-butene, and 2,3-dimethyl-1-butene. Draw the structure of the carbocation intermediate leading to the formation of 2,3-dimethyl-2-butene. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • If more than one structure fits the description, draw them all. • Separate structures with + signs from the drop-down menu. / H₂C CH₂ CH3 CH3 ChemDoodleⓇ On []arrow_forwardAcid-catalyzed dehydration of 3,3-dimethyl-2-butanol gives three alkenes: 2,3-dimethyl-2-butene, 3,3-dimethyl-1-butene, and 2,3-dimethyl-1- butene. Draw the structure of the carbocation intermediate leading to the formation of 2,3-dimethyl-2-butene. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. If more than one structure fits the description, draw them all. Separate structures with + signs from the drop-down menu. C P opy aste ChemDoodle >arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License