
Concept explainers
Interpretation:
The mechanism for each step in the pathway given for bio synthesizing epi-Aristolochene is to be shown using curved arrows. Further the steps that involve electrophilic addition(s) to the
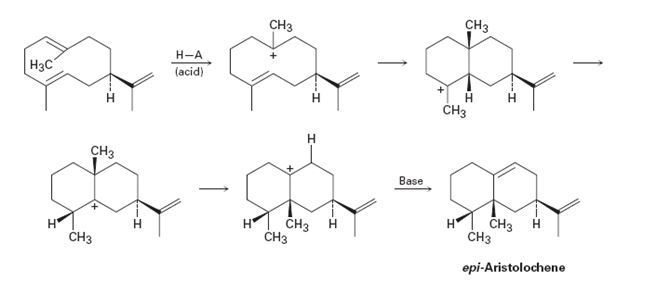
Concept introduction:
While drawing curved arrows the head of the curved arrow starts from the nucleophilic source (either negatively charged or neutral) and ends in an electrophilic sink (either positively charged or neutral). The formation of a new bond should lead to the breaking of a bond that already exists as the octet rule cannot be violated.
In electrophilic addition reactions, the first step is the attack of the π electrons of the double bond on the hydrogen of another reactant to yield a carbocation. One of the carbon in C=C gets attached to hydrogen while the other acquires a positive charge. In the second step, the carbocation formed can rearrange to give another more stable carbocation either by a hydride shift (shift of hydrogen atom with its electron pair) or by an alkyl shift (shift of an alkyl group with its electron pair) between neighboring carbons. In the last step the carbocation produced reacts with a negatively charged ion or eliminates a proton to give the product.
To show:
The mechanism for each step in the pathway given for bio synthesizing epi-aristolochene using curved arrows. Further to identify the steps that involve electrophilicaddition(s) to the alkene and that involve carbocation rearrangement(s).
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry
- A proton (H*) from trifluoromethanesulfonic acid, CF3SO2OH, can add to the alkyne shown to yield two different carbocation products. (a) Draw the mechanism for each of these steps, along with the corresponding products. (b) Which carbocation is more stable? H3C C-c=CH H2 + H-OSO,CFз ? H2arrow_forwardIdentify the major organic product and provide a plausible mechanism (curved arrows) for the following reaction. In your mechanism, you should include any resonance structures that may help to stabilise the reaction intermediate. CI ZI MeCN (Solvent) ?arrow_forwardDraw the mechanism for the reaction of an alkyl halide with sodium azide followed by reduction. Complete the mechanism of the initial step of the reaction, then identify the key intermediate and the product. Step 1: Draw curved arrows. o z + Na + || : z: I Step 2: Complete the intermediate. Na +arrow_forward
- In the presence of light or heat, diazomethane (CH,N2) creates a carbene, which then adds to an alkene to make a cyclopropane. In the boxes below. draw the mechanism arrows for the reaction. H INEN: INE N:arrow_forwardThe rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form glycylglycine ethyl ester is 0.6 M-1s-1. In the presence of Co2+, the rate constant is 1.5 x 106 M-1s-1. What rate enhancement does the catalyst provide?arrow_forwardA carbamate can be prepared by treating an isocyanate with an alcohol, as shown here. This type of reaction is used to synthesize polyurethanes- polymers that have a wide variety of industrial applications, such as surface sealants, high-performance adhesives, and synthetic fibers. Propose a mechanism for this transformation. R'—ОН R. `N=C=0 R. OR' An isocyanate A carbamate (Substituted urethane) O=Carrow_forward
- Provide the mechanism for the following reaction (1) PhMgBr (2 equiv) (2) H3O+ workup OHarrow_forward(c) The following reaction shows the electrophilic addition reaction between an alkene compound with hydrogen chloride, HCI. CI + HCI Major product Draw the mechanism for the formation of major product.arrow_forwardu) Show the mechanism for the following Elimination Reactions. Х I Но у N 1 HI + H2Oarrow_forward

 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

