
Concept explainers
a)
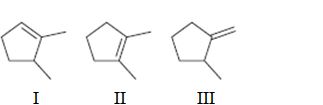
Interpretation:
To rank the double bond in the compounds given in terms of increasing stability.
Concept introduction:
The stability of a double bond can be determined by the number of hydrogens on adjacent carbons attached to both carbons in the double bond. (H’s on the α carbon). More the number of such hydrogens, the more hyperconjugation occur and more stable the
b)
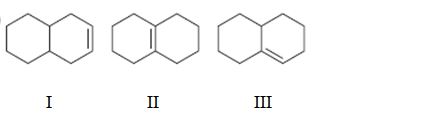
Interpretation:
To rank the double bond in the compounds given in terms of increasing stability.
Concept introduction:
The stability of a double bond can be determined by the number of hydrogens on adjacent carbons attached to both carbons in the double bond.(H’s on the α carbon). More the number of such hydrogens, the more hyperconjugation occur and more stable the alkene.
c)
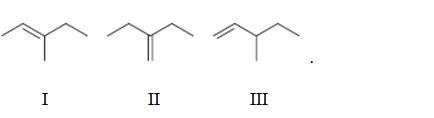
Interpretation:
To rank the double bond in the compounds given in terms of increasing stability.
Concept introduction:
The stability of a double bond can be determined by the number of hydrogens on adjacent carbons attached to both carbons in the double bond.(H’s on the α carbon). More the number of such hydrogens, the more hyperconjugation occur and more stable the alkene.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry
- Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic steps. Be sure to account for all bond-breaking and bond-making steps. :OH: Select to Add Arrows H₂O 3 H₂ H HH H :Br:Ⓒ HH Select to Add Arrows H₂O 17 :Br: H H Please select a drawing or reagent from the question areaarrow_forwardRank the following according to INCREASING reactivity towards SN2: 1st (least reactive)? 2ND? 3RD? 4TH (most reactive)?arrow_forwardWhich compound has conjugated C=C bonds? А. С. D. B.arrow_forward
- Which indicated bond in each pair of compounds has the higher bond dissociation energy? Which is the stronger bond?arrow_forwardRank the free radicals shown below in order of increasing stability:arrow_forwardWhich of the following resonance structures is the greatest contributor to the resonance hybrid?arrow_forward
- Curved arrows are used to illustrate the flow of electrons. Using the provided resonance structures, draw the curved electron- pushing arrows to show the interconversion between resonance hybrid contributors. Be sure to account for all bond- breaking and bond-making steps. I I I I :O: farrow_forwardCan you rank these from most to least stable? thank you!arrow_forwardArrange the following radicals in order of increasing stability. A B C D Earrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

